Imagine an electron in the 5f orbital of a neutral atom. Decide how well each of...
Fantastic news! We've Found the answer you've been seeking!
Question:
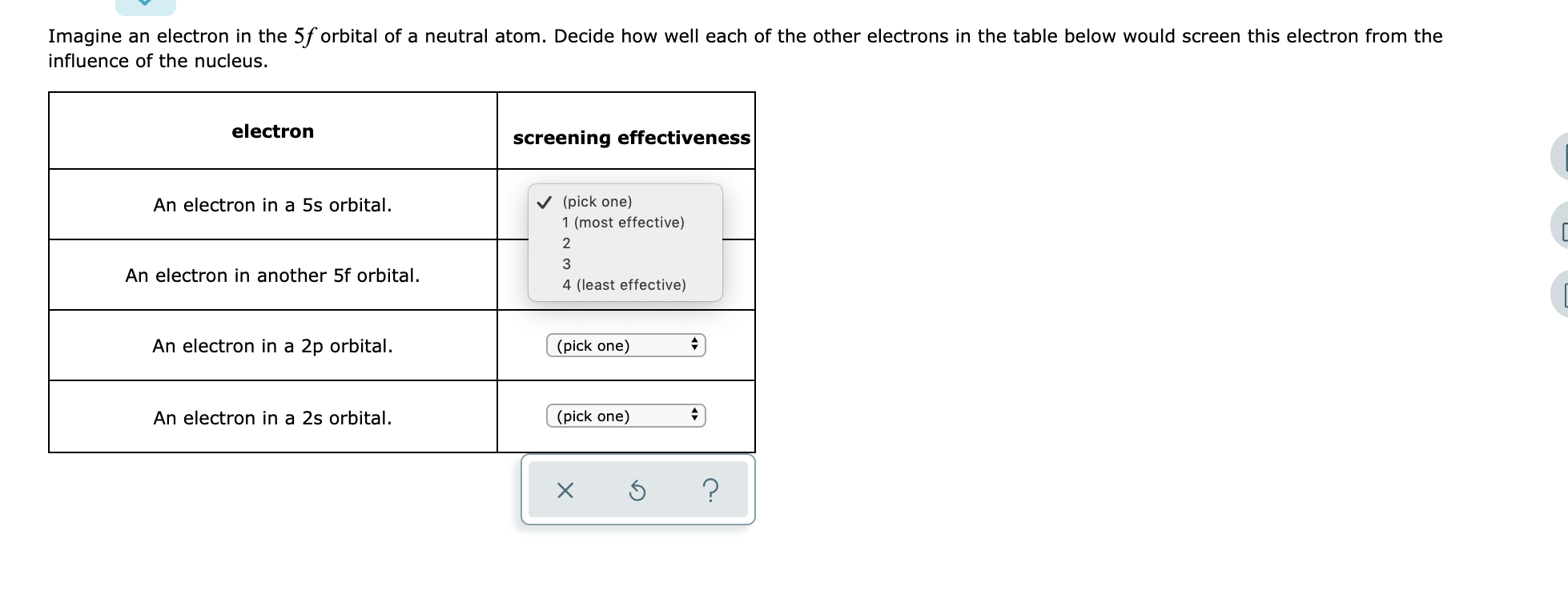
Transcribed Image Text:
Imagine an electron in the 5f orbital of a neutral atom. Decide how well each of the other electrons in the table below would screen this electron from the influence of the nucleus. electron An electron in a 5s orbital. An electron in another 5f orbital. An electron in a 2p orbital. An electron in a 2s orbital. screening effectiveness ✓ (pick one) 1 (most effective) 2 3 4 (least effective) (pick one) (pick one) X + ♦ ? Imagine an electron in the 5f orbital of a neutral atom. Decide how well each of the other electrons in the table below would screen this electron from the influence of the nucleus. electron An electron in a 5s orbital. An electron in another 5f orbital. An electron in a 2p orbital. An electron in a 2s orbital. screening effectiveness ✓ (pick one) 1 (most effective) 2 3 4 (least effective) (pick one) (pick one) X + ♦ ?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The electron configuration of a neutral atom is 1s22s22p63s2. Write a complete set of quantum numbers for each of the electrons. Name the element.
-
An excited hydrogen atom with an electron in the n = 5 state emits light having a frequency of 6.90 1014 s-1. Determine the principal quantum level for the final state in this electronic transition.
-
A hydrogen atom has an electron in the n = 5 level. (a) If the electron returns to the ground state by emitting radiation, what is the minimum number of photons that can be emitted? (b) What is the...
-
Problem 5: Closing entries. (10 points) Given the following accounts, prepare the closing entries in the order and method taught using Income Summary. Number each entry 1, 2, .... Accounts payable $...
-
A partner in a medium-size A/E (architectural/engineering) design firm is evaluating two alternatives for improving the exterior appearance of the building they occupy. The building can be completely...
-
An FHSS system uses a 4-bit PN sequence. If the bit rate of the PN is 64 bits per second, answer the following questions: a. What is the total number of possible channels? b. What is the time needed...
-
Assume that Fossil paid expenses totaling \($40,000\). How does this transaction affect Fossils accounting equation? a. Increases assets and decreases liabilities b. Decreases assets and increases...
-
The Commonwealth of Massachusetts has passed a new law which requires all Medical Health Professionals, (hereinafter MHP) which includes but is not limited to medical doctors, psychiatrists, social...
-
The truss is loaded by a vertical point force F . The rods of the truss have the same Young's modulus E and the second moment of area I . The cross section of the rod is a circle. Determine the rod...
-
Kat Ltd.s September balance sheet contains the following information: Cash.............................................................................$ 37,500 (dr) Accounts...
-
A solution of HNO, is standardized by reaction with pure sodium carbonate. 2H+ + NaCO, 2 Na+ + HO + CO A volume of 23.28 0.05 mL of HNO, solution was required for complete reaction with 0.8463...
-
A firm reported year-end sales of $20 million. It listed $7 million of inventory on its balance sheet. Using a 365-day year, how many days did the firm's inventory stay on the premises?
-
An investment offers the following cash flows: $554 today, $182 one year from now, $482 in 2 years, and $793 in 3 years. If the relevant interest rate is 7% per year (an APR, with interest compounded...
-
A baseball is hit when it is 3 feet above the ground. It leaves the bat with initial 152ft speed of making an angle of 20 with the horizontal. At the instant the ball is sec hit, an instantaneous...
-
For a Proposed Infinite Google Tablet 3.3 Objectives Set specific marketing objectives. Include Objectives that correlate to: Product Category market share, Specific target market share, Market...
-
What is the internal rate of return (IRR) for a project that costs $5,500 and is expected to generate $1,800 per year for the next four years? If the firm's required rate of return is 8 percent, what...
-
Question 15 of 40 Some information can be communicated through charts. To share information about a project's timeline, which chart would a project manager most likely use? O a P31 chart a Pareto...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
Although the VSEPR model is correct in predicting that CH4 is tetrahedral, NH3 is pyramidal, and H2O is bent, the model in its simplest form does not account for the fact that these molecules do not...
-
The following are representations of acid base reactions: a. Label each of the species in both equations as an acid or a base and explain. b. For those species that are acids, which labels apply:...
-
A sample of chloroform is found to contain 12.0 g of carbon, 106.4 g of chlorine, and 1.01 g of hydrogen. If a second sample of chloroform is found to contain 30.0 g of carbon, what is the total mass...
-
Distinguish among the following tests and indicate the assertions to which each test pertains: a. Vouch recorded payables to supporting documentation. b. Examine subsequent payments. c. Determine...
-
a. Discuss materiality from the perspective of the production cycle. b. Discuss inherent risk from the perspective of the production cycle. c. Why might the auditor use different audit strategies for...
-
What circumstances pertaining to property, plant, and equipment may warrant the following assessments of inherent risk (IR)? a. Low for the existence or occurrence assertion. b. Moderate or high for...

Study smarter with the SolutionInn App



