Imported ceramic bowls are analyzed for toxic heavy metals, such as cadmium, because these metals can...
Fantastic news! We've Found the answer you've been seeking!
Question:
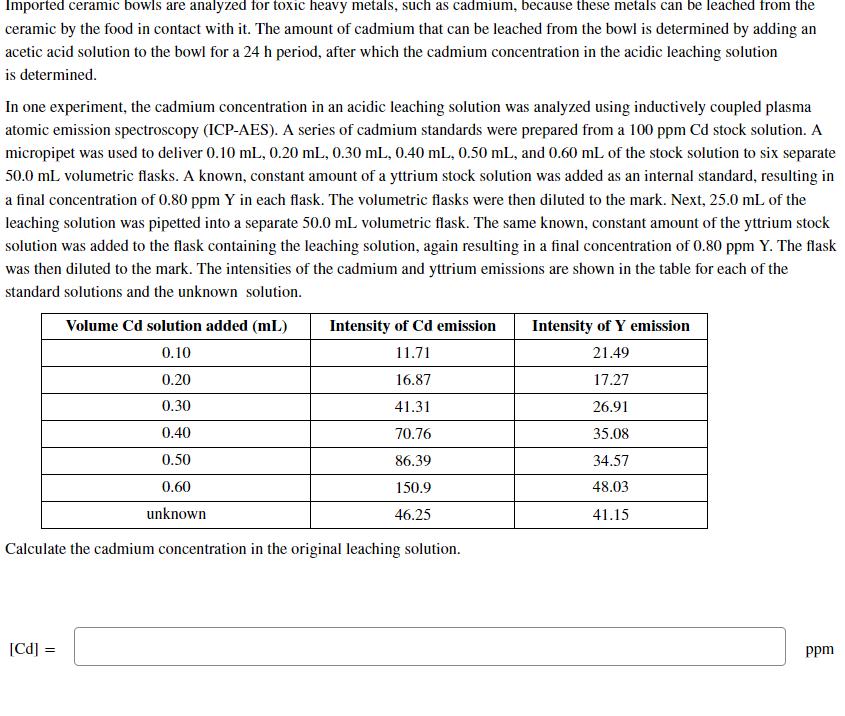
Transcribed Image Text:
Imported ceramic bowls are analyzed for toxic heavy metals, such as cadmium, because these metals can be leached from the ceramic by the food in contact with it. The amount of cadmium that can be leached from the bowl is determined by adding an acetic acid solution to the bowl for a 24 h period, after which the cadmium concentration in the acidic leaching solution is determined. In one experiment, the cadmium concentration in an acidic leaching solution was analyzed using inductively coupled plasma atomic emission spectroscopy (ICP-AES). A series of cadmium standards were prepared from a 100 ppm Cd stock solution. A micropipet was used to deliver 0.10 mL, 0.20 mL, 0.30 mL, 0.40 mL, 0.50 mL, and 0.60 mL of the stock solution to six separate 50.0 mL volumetric flasks. A known, constant amount of a yttrium stock solution was added as an internal standard, resulting in a final concentration of 0.80 ppm Y in each flask. The volumetric flasks were then diluted to the mark. Next, 25.0 mL of the leaching solution was pipetted into a separate 50.0 mL volumetric flask. The same known, constant amount of the yttrium stock solution was added to the flask containing the leaching solution, again resulting in a final concentration of 0.80 ppm Y. The flask was then diluted to the mark. The intensities of the cadmium and yttrium emissions are shown in the table for each of the standard solutions and the unknown solution. Volume Cd solution added (mL) 0.10 0.20 0.30 0.40 0.50 0.60 unknown [Cd] = Intensity of Cd emission 11.71 16.87 41.31 70.76 86.39 150.9 46.25 Calculate the cadmium concentration in the original leaching solution. Intensity of Y emission 21.49 17.27 26.91 35.08 34.57 48.03 41.15 ppm Imported ceramic bowls are analyzed for toxic heavy metals, such as cadmium, because these metals can be leached from the ceramic by the food in contact with it. The amount of cadmium that can be leached from the bowl is determined by adding an acetic acid solution to the bowl for a 24 h period, after which the cadmium concentration in the acidic leaching solution is determined. In one experiment, the cadmium concentration in an acidic leaching solution was analyzed using inductively coupled plasma atomic emission spectroscopy (ICP-AES). A series of cadmium standards were prepared from a 100 ppm Cd stock solution. A micropipet was used to deliver 0.10 mL, 0.20 mL, 0.30 mL, 0.40 mL, 0.50 mL, and 0.60 mL of the stock solution to six separate 50.0 mL volumetric flasks. A known, constant amount of a yttrium stock solution was added as an internal standard, resulting in a final concentration of 0.80 ppm Y in each flask. The volumetric flasks were then diluted to the mark. Next, 25.0 mL of the leaching solution was pipetted into a separate 50.0 mL volumetric flask. The same known, constant amount of the yttrium stock solution was added to the flask containing the leaching solution, again resulting in a final concentration of 0.80 ppm Y. The flask was then diluted to the mark. The intensities of the cadmium and yttrium emissions are shown in the table for each of the standard solutions and the unknown solution. Volume Cd solution added (mL) 0.10 0.20 0.30 0.40 0.50 0.60 unknown [Cd] = Intensity of Cd emission 11.71 16.87 41.31 70.76 86.39 150.9 46.25 Calculate the cadmium concentration in the original leaching solution. Intensity of Y emission 21.49 17.27 26.91 35.08 34.57 48.03 41.15 ppm
Expert Answer:

Related Book For 

An Introduction To Statistical Methods And Data Analysis
ISBN: 9781305465527
7th Edition
Authors: R. Lyman Ott, Micheal T. Longnecker
Posted Date:
Students also viewed these economics questions
-
The EPA set emission standards for vinyl chloride, a toxic substance that is carcinogenic to humans. The Clean Air Act says such standards must be "at the level which.provides an ample margin of...
-
AES was formed in 1996, at which time it hired eight employees. At a meeting of these employees in 1997, they expressed concern that the company might not survive, as it was using outdated equipment....
-
Acetic acid can be manufactured by combining methanol with carbon monoxide, an example of a carbonylation reaction: (a) Calculate the equilibrium constant for the reaction at 25°C. (b)...
-
____, one-way ANOVA compares the means of three or more independent samples.
-
Cite examples of expenses you believe are commonly incurred in integrating target companies. Be specific.
-
Two jobs were completed with total costs of $183,000 and $105,000 respectively. They were sold on account at a mark-up of 50% on cost. After completing these transactions, the balance in the Cost of...
-
What are the characteristics of business domains? Explain them in the context of a specific business.
-
Macon Publishing House produces consumer magazines. The house and home division, which sells home-improvement and home-decorating magazines, has seen a 20% reduction in operating income over the past...
-
3. Suppose a local post office requires the following number of workers on the different days of the week. Day Number of Day workers Number of workers Mon 11 Fri 20 Tue 12 Sat Wed Thu 13 14 Sun 10 10...
-
Consider the deterministic world below (part (a)). Allowable moves are shown by arrows, and the numbers indicate the reward for performing each action. If there is no number, the reward is zero....
-
Which of the following is true? (a) If p is prime and p l ab then p la and plb (b) If a|c and b|c, then (a + b)|c (c) There are infinitely many primes of the form n 9. (d) If a|b and a|c, then a|(bc)
-
Visit the website, Kernel Newbies (https://kernelnewbies.org/LinuxChanges), and examine the latest version of the Linux kernel, as well as the major hardware and software features that were added in...
-
The SEC 10-K assignment will consist of research and the completion of a paper on select areas of an annual SEC 10-K report filed by a public company with the U.S. Securities and Exchange Commission...
-
(d) Write a function named void readDurianDetails (Durian durian[], int &noDurian) that read the information from the text file of durianDetails.txt and display the types of durians with the taste...
-
Pablo and Jacob are running a half marathon. Jacob is d = 40.1 m behind Pablo, and both are running at the same speed, Vo = 2.53 m/s. At a particular moment, Jacob begins to accelerate forward at a...
-
Find the value of an investment of $20,000 for 13 years at an annual interest rate of 3.85% compounded continuously The value of the investment is $?
-
What is the Comprehensive Plan to Reduce the Incidence of Lung Cancer within the Smoking Community
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
In the January 2004 issue of Consumer Reports, an article titled "Cut the Fat" described some of the possible problems in the diets of the U. S. public. The following table gives data on the increase...
-
Defining the Problem (1). Lead is an environmental pollutant especially worthy of attention because of its damaging effects on the neurological and intellectual development of children. Morton et al....
-
Refer to Exercise 14.8. a. Draw a profile plot for the two factors, age and product type. b. Perform appropriate F tests and draw conclusions from these tests concerning the effects of age and...
-
Jennifer knows that Carters malefemale differential is indefensible? What should she do to rectify it?
-
Is there another incentive plan you think would work better for the pressers? Describe it.
-
A store managers job is to keep total wages to no more than 30% of sales and to maintain the fuel bill and the supply bill at about 9% of sales each. Managers can also directly affect sales by...

Study smarter with the SolutionInn App


