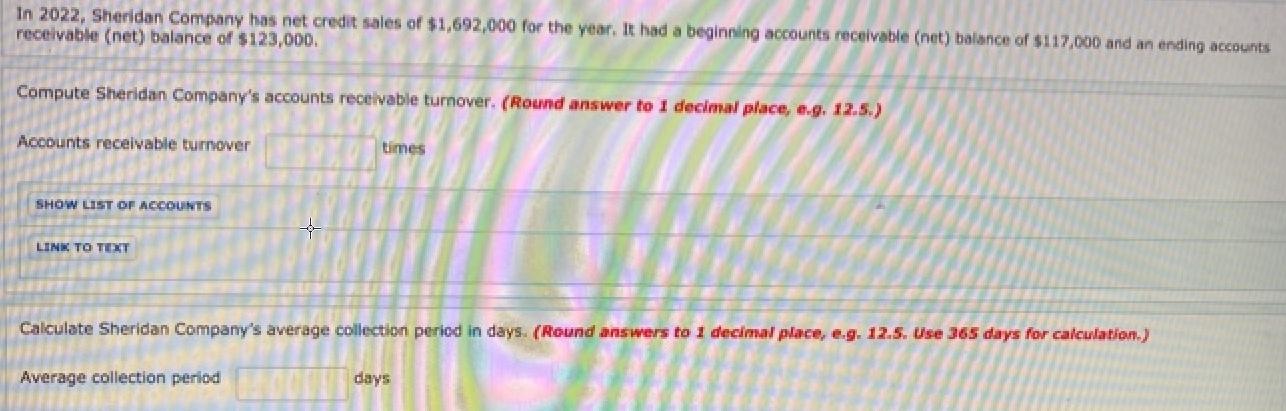
In 2022, Sheridan Company has net credit sales of $1,692,000 for the year. It had a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In 2022, Sheridan Company has net credit sales of $1,692,000 for the year. It had a beginning accounts receivable (net) balance of $117,000 and an ending accounts receivable (net) balance of $123,000. Compute Sheridan Company's accounts receivable turnover. (Round answer to 1 decimal place, e.g. 12.5.) Accounts receivable turnover times SHOW LIST OF ACCOUNTS + LINK TO TEXT Calculate Sheridan Company's average collection period in days. (Round answers to 1 decimal place, e.g. 12.5. Use 365 days for calculation.) Average collection period days In 2022, Sheridan Company has net credit sales of $1,692,000 for the year. It had a beginning accounts receivable (net) balance of $117,000 and an ending accounts receivable (net) balance of $123,000. Compute Sheridan Company's accounts receivable turnover. (Round answer to 1 decimal place, e.g. 12.5.) Accounts receivable turnover times SHOW LIST OF ACCOUNTS + LINK TO TEXT Calculate Sheridan Company's average collection period in days. (Round answers to 1 decimal place, e.g. 12.5. Use 365 days for calculation.) Average collection period days
Expert Answer:
Answer rating: 100% (QA)
Accounts Recievable Turn over ratio Numerator Denominator Accou... View the full answer

Related Book For 

Financial and managerial accounting
ISBN: 978-1118016114
1st edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Posted Date:
Students also viewed these accounting questions
-
In 2014, Lauren Company has net credit sales of $1,480,000 for the year. It had a beginning accounts receivable (net) balance of $112,000 and an ending accounts receivable (net) balance of $108,000....
-
In 2014, Wainwright Company has net credit sales of $1,300,000 for the year. It had a beginning accounts receivable (net) balance of $101,000 and an ending accounts receivable (net) balance of...
-
In 2017, Wainwright Company has net credit sales of $1,300,000 for the year. It had a beginning accounts receivable (net) balance of $101,000 and an ending accounts receivable (net) balance of...
-
The current quoted price of a 13% coupon bond is $110. It pays coupon semi-annually. The next coupon will be paid in 6-days (total number of days in this semi-annual period is 181) and the futures...
-
CPA Kara Rambo is the auditor of Ajax Corporation. Her audit independence will not be considered impaired if she a. Owns $ 1,000 worth of Ajax stock. b. Has a husband who owns $ 1,000 worth of Ajax...
-
Post the following transactions to the ledger of Kingston Company. The partial ledger of Kingston Company includes Cash, 111; Equipment, 121; Accounts Payable, 211; and A. Kingston, Capital, 311....
-
Calculate the heating season auxiliary energy required for the Boston house in Problem 9.8. Problem 9.8 Using the SLR method, calculate the auxiliary energy required in March for a \(2000...
-
Robert Berry is the managing partner for Berry & Associates (B&A), LLP. He was recently reviewing the firms income statement for the previous quarter, which showed that auditing revenues were about 5...
-
Samuels Manufacturing is considering the purchase of a new machine to replace one it believes is obsolete.The firm has total current assets of $930,000 and total current liabilities of $644,000. As a...
-
Conch Republic Electronics is a midsized electronics manufacturer located in Key West, Florida. The company president is Shelley Couts, who inherited the company. When it was founded over 70 years...
-
How do organizations measure the impact and ROI of their organizational development initiatives, utilizing a combination of qualitative and quantitative metrics to assess effectiveness, drive...
-
What is the purpose of adding coatings to cutting tool materials? a ) ) To increase tool hardness b ) ) To improve thermal conductivity c ) ) To enhance wear resistance d ) ) To reduce tool vibration
-
A Boeing 737 airliner has a mass of 20,000 kg and the total area of both wings (top or bottom) is 100 m. What is the pressure difference between the top and bottom surface of each wing when the...
-
5- The lift slope for an airfoil is 0.1123 degree-, and az-o = -0.8. Consider a finite wing using this airfoil, with AR = 6 and taper ratio = 0.8. Assume that 6 = T. Calculate the lift coefficient...
-
if your fingers apply a couple Considering the drill chuck where: F = 2.5lb (in both cases) and d = 2.2 in, what will be the magnitude of the moment 'M' tightening the drill if the chuck key gear...
-
4- Consider an airplane. For this airplane the zero-lift angle of attack is -1.5, the lift slope (ao) of the airfoil section is 0.112 per degree, the lift efficiency factor t = 0.04, and the wing...
-
Make a frequency table and relative frequency table of the data. A B AB AO O 0 AB B 1. Individual: 4. 2. Variable: B B Blood Type A B AB 0 B A O O O AB A AB 3. Type of variable: Qualitative (circle...
-
Use Stokes' Theorem to evaluate f(y+sin x) dx+(z+cos y) dy+rdz, where C is the rve r(t) = (sint, cost, sin 2t), t = [0, 2].
-
Tempo Ltd. is a retailer operating in Dartmouth, Nova Scotia. Tempo uses the perpetual inventory method. All sales returns from customers result in the goods being returned to inventory; the...
-
On November 1, 2014, the account balances of Samone Equipment Repair, Inc. were as follows. During November, the following summary transactions were completed. Nov. 8 Paid $1,500 for salaries due...
-
Peine Candle Supply makes candles. The sales mix (as a percentage of total dollar sales) of its three product lines is birthday candles 30%, standard tapered candles 50%, and large scented candles...
-
A system was prepared by partially decomposing \(\mathrm{CaCO}_{3}\) into an evacuated space. What is the number of degrees of freedom \((f)\) for the system?
-
Prove that chemical potentials of two phases in equilibrium are equal.
-
Derive the following relation: \[ \left(\frac{\partial C_{V}}{\partial V} ight)_{T}=T\left(\frac{\partial^{2} P}{\partial T^{2}} ight)_{V} \]

Study smarter with the SolutionInn App


