LE3AF 600 N 1 2 4 5 7 8 9 10 11 12 13 14 15...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
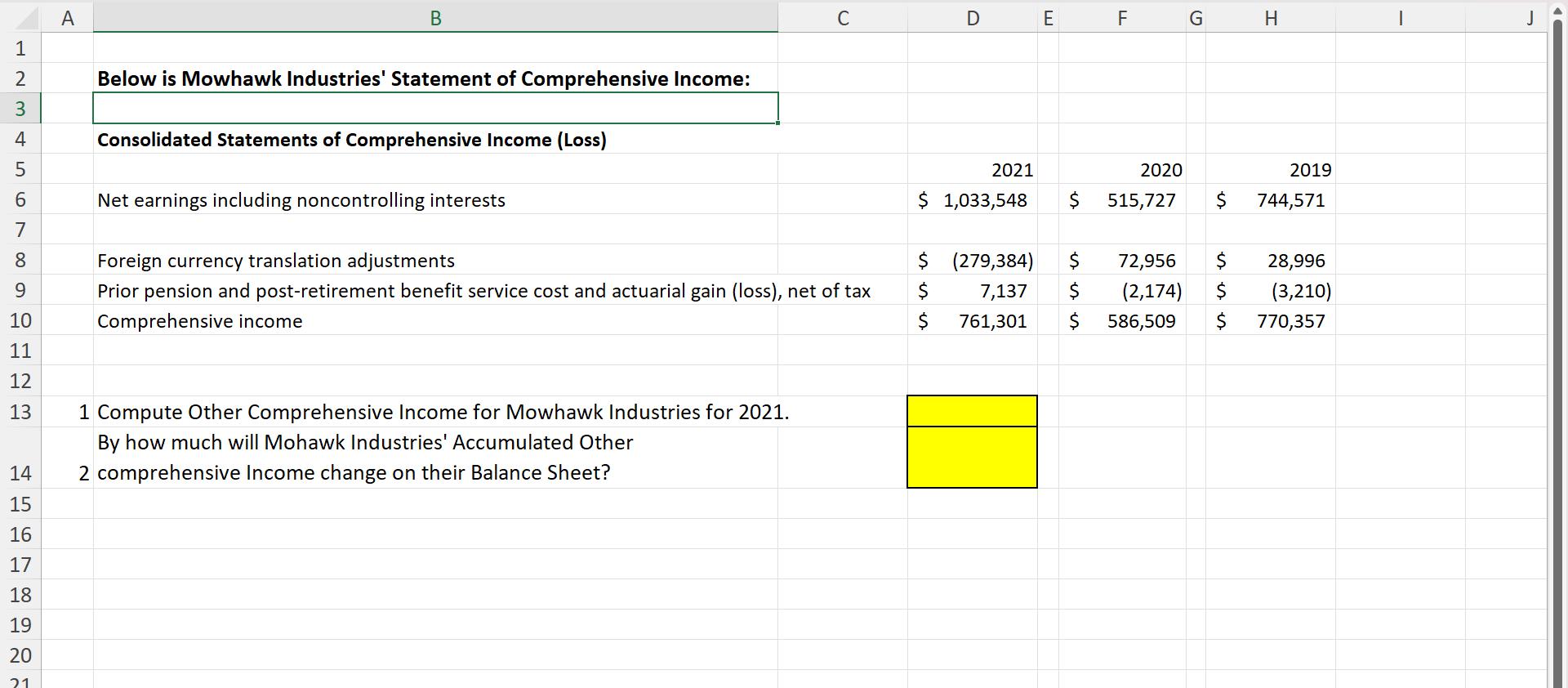
LE3AF 600 N 1 2 4 5 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 A B Below is Mowhawk Industries' Statement of Comprehensive Income: Consolidated Statements of Comprehensive Income (Loss) Net earnings including noncontrolling interests C Foreign currency translation adjustments Prior pension and post-retirement benefit service cost and actuarial gain (loss), net of tax Comprehensive income 1 Compute Other Comprehensive Income for Mowhawk Industries for 2021. By how much will Mohawk Industries' Accumulated Other 2 comprehensive Income change on their Balance Sheet? D 2021 $ 1,033,548 $ $ $ (279,384) 7,137 761,301 E F $ 2020 $ 515,727 $ G 72,956 $ $ (2,174) 586,509 H 2019 744,571 28,996 (3,210) $ 770,357 I D LE3AF 600 N 1 2 4 5 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 A B Below is Mowhawk Industries' Statement of Comprehensive Income: Consolidated Statements of Comprehensive Income (Loss) Net earnings including noncontrolling interests C Foreign currency translation adjustments Prior pension and post-retirement benefit service cost and actuarial gain (loss), net of tax Comprehensive income 1 Compute Other Comprehensive Income for Mowhawk Industries for 2021. By how much will Mohawk Industries' Accumulated Other 2 comprehensive Income change on their Balance Sheet? D 2021 $ 1,033,548 $ $ $ (279,384) 7,137 761,301 E F $ 2020 $ 515,727 $ G 72,956 $ $ (2,174) 586,509 H 2019 744,571 28,996 (3,210) $ 770,357 I D
Expert Answer:
Answer rating: 100% (QA)
To compute the Other Comprehensive Income for Mohawk Industries for the year 2021 we need to add the ... View the full answer

Related Book For 

Introduction to Operations Research
ISBN: 978-1259162985
10th edition
Authors: Frederick S. Hillier, Gerald J. Lieberman
Posted Date:
Students also viewed these finance questions
-
Participate in a discussion with your classmates regarding economic reasoning in government policies. Recall that economic reasoning is based on the premise that everything has a cost, thus, economic...
-
Monthly payments were originally calculated to repay a $20,000 loan at 9% compounded monthly over a 10-year period. After one year, the debtor took advantage of an option in the loan contract to...
-
How much can a car buyer finance with a 5 year loan Payments are monthly and they start at the beginning of the month The loan requires no downpayment Monthly payment is 540 rate of interst is 7
-
On July 1, Novak Corporation purchases 570 shares of its $5 par value common stock for the treasury at a cash price of $8 per share. On September 1, it sells 320 shares of the treasury stock for cash...
-
What are the benefits of using simulation to evaluate capital investment projects?
-
Identify the major organic product of each of the following reactions. If two regioisomers are formed in appreciable amounts, show them both. (a) (b) (c) Product of part (b) + dilute hydrochloric...
-
Fugacity and pressure are numerically not equal for a gas (a) At low temperature and high pressure (b) At standard state (c) Both (a) and (b) (d) In an ideal state.
-
Cash Dividends Kerry Company has 1,000 shares of $100 par value, 9% preferred stock and 10,000 shares of $10 par value common stock outstanding. The preferred stock is cumulative and...
-
During 2024, a company sells 389 units of inventory for $94 each. The company has the following inventory purchase transactions for 2024: Number of Unit Date January 1 May 5 Transaction Beginning...
-
Toronto Cricket Property Inc. (TPI) has been incorporated with the purpose of build and operate a world-class cricket stadium outside Toronto. The land on which the stadium will be build is currently...
-
Let (X,B) be a t-(v, k, ) design. (a) For 0 < s
-
You are a bookkeeper at a medium-sized manufacturing company called XYZ Manufacturing Pty Ltd. The company has been growing steadily over the past few years, and as a result, the number of fixed...
-
Zinc Limited (ZL) has entered into the following transactions: (i) On 1 January 2022, ZL purchased 1.5 million bonds of Copper Limited having face value of Rs. 100 each at a premium of Rs. 5 each...
-
Q1- Operating Budget (15 points) You have been asked by your VP of Finance to put together the Operating Budgets and the Projected Income Statement for Comfy Bedding Co, using the information...
-
Choose the correct statement to initialize a loop in the below code: newList=["Hello", 1, True, 4.5] print(listitem) for listitem in newList: for newList in listItem: for listltem in newlist: for...
-
What will display when the following code is executed? Do not worry about line breaks as Canvas does not allow me to include line breaks in answers. x = range(2, 7, 2) for n in x: print(n, "ghosts")
-
You have just graduated from university and are considering starting a new line clothing. To start the new line of clothing you require a capital outlay of $25,000 and net working capital of $1,000....
-
Show that the block upper triangular matrix A in Example 5 is invertible if and only if both A 11 and A 22 are invertible. Data from in Example 5 EXAMPLE 5 A matrix of the form A = [ A11 A12 0 A22 is...
-
Suppose that the three-variable linear programming problem given in Fig. 5.2 has the objective function Maximize Z = 3x1 + 4x2 + 3x3. Without using the algebra of the simplex method, apply just its...
-
Reconsider the Union Airways problem and its spreadsheet model that was dealt with in Prob. 7.3-6. Management now is considering increasing the level of service provided to customers by increasing...
-
Consider the discrete random variable X that is uniformly distributed (equal probabilities) on the set {1, 2, . . . , 9}. You wish to generate a series of random observations xi (i = 1, 2, . . .) of...
-
The freezing point of pure benzene is \(5.44^{\circ} \mathrm{C}\) and that of the solution containing \(2.092 \mathrm{~g}\) of benzaldehyde in \(100 \mathrm{~g}\) of benzene is \(4.44^{\circ}...
-
The molality of dissolved gases in water at \(0^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\) is \(1.29 \times 10^{-3}\). The decrease in volume during melting of ice is \(0.0907 \mathrm{cc} /...
-
Calculate the mass of methyl alcohol which, when dissolved in \(100 \mathrm{~g}\) of water, would just prevent the formation of ice at \(-10^{\circ} \mathrm{C}\), given that \(K_{\mathrm{f}}\) is...

Study smarter with the SolutionInn App


