Question: Why is it impossible to perform a liquid-liquid extraction using HO and CHOH as the two solvents? 2 Suppose a mixture of two immiscible
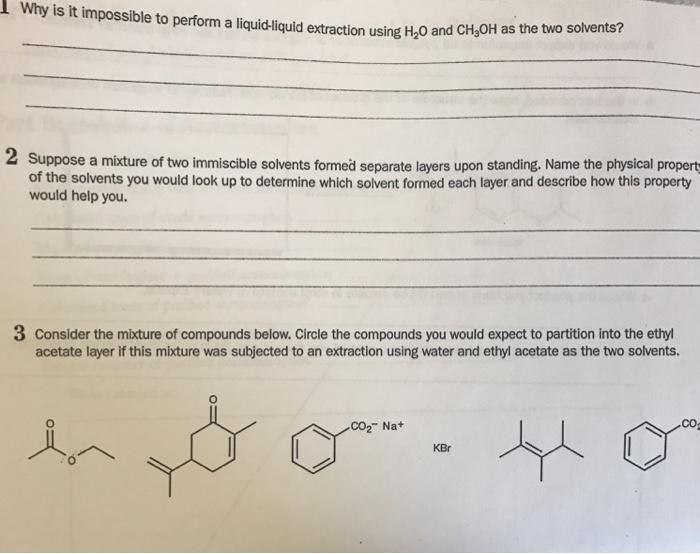
Why is it impossible to perform a liquid-liquid extraction using HO and CHOH as the two solvents? 2 Suppose a mixture of two immiscible solvents formed separate layers upon standing. Name the physical propert of the solvents you would look up to determine which solvent formed each layer and describe how this property would help you. 3 Consider the mixture of compounds below. Circle the compounds you would expect to partition into the ethyl acetate layer if this mixture was subjected to an extraction using water and ethyl acetate as the two solvents. lon CO Na+ KBr
Step by Step Solution
There are 3 Steps involved in it
1 LiquidLiquid extraction refers to or separating drawing compound o... View full answer

Get step-by-step solutions from verified subject matter experts


