In a nuclear incident, for 10 hours, a nuclear worker with a mass of (70+ LDSN)...
Fantastic news! We've Found the answer you've been seeking!
Question:
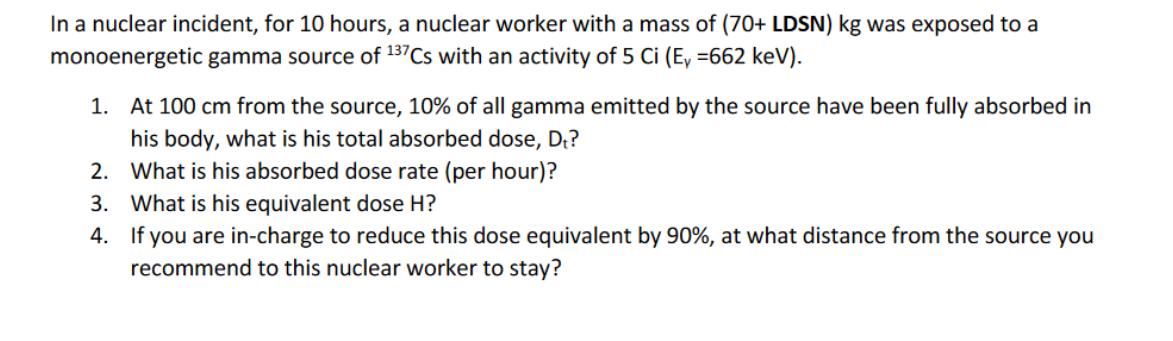
Transcribed Image Text:
In a nuclear incident, for 10 hours, a nuclear worker with a mass of (70+ LDSN) kg was exposed to a monoenergetic gamma source of 137Cs with an activity of 5 Ci (E, =662 keV). 1. At 100 cm from the source, 10% of all gamma emitted by the source have been fully absorbed in his body, what is his total absorbed dose, D₁? 2. What is his absorbed dose rate (per hour)? 3. What is his equivalent dose H? 4. If you are in-charge to reduce this dose equivalent by 90%, at what distance from the source you recommend to this nuclear worker to stay? In a nuclear incident, for 10 hours, a nuclear worker with a mass of (70+ LDSN) kg was exposed to a monoenergetic gamma source of 137Cs with an activity of 5 Ci (E, =662 keV). 1. At 100 cm from the source, 10% of all gamma emitted by the source have been fully absorbed in his body, what is his total absorbed dose, D₁? 2. What is his absorbed dose rate (per hour)? 3. What is his equivalent dose H? 4. If you are in-charge to reduce this dose equivalent by 90%, at what distance from the source you recommend to this nuclear worker to stay?
Expert Answer:
Answer rating: 100% (QA)
C5Ci E662 KeV m70kg t10hrs 1 Total absorbed dose 01DRtimeequvalent ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
An inclined manometer is a useful device for measuring small pressure differences. The formula given in Section 3.4 for the pressure difference in terms of the liquid-level difference h remains...
-
What is happening to world markets, and what are the implications for financial reporting?
-
Suppose the average price of electricity for a New England customer follows the continuous uniform distribution with a lower bound of 12 cents per kilowatt-hour and an upper bound of 20 cents per...
-
The dataset ToyotaCorolla.jmp contains data on used cars on sale during the late summer of 2004 in the Netherlands. It has 1436 records containing details on 38 attributes, including Price, Age,...
-
Hovington, CPA, knows that while audit objectives relating to inventories may be stated in terms of the assertions as presented in this chapter, they may also be subdivided and stated more...
-
Mrs Kebede S, consumes two good A and B, when Ke bede,s income increase from birr 4000 to 5000, the quantity of good A demanded by increase Kebede e by 35%. What conclusion do you draw from this.
-
At the end of March, Weber Productions accounting records reveal a balance for cash equal to $21,861. However, the balance of cash in the bank at the end of February is only $4,576. Weber is...
-
a) (4 marks) ABC LTD's's ordinary shares are currently selling for R218.40 per share, with a current dividend (Do) of R12.88 per share. The firm's dividends have been growing at an annual rate of...
-
A polling organization conducts a study to estimate the percentage of households that have both parents sharing equally in household chores. It mails a questionnaire to 1228 randomly selected...
-
To help assess student learning in her music theory courses, a music professor at a community college implemented pre- and post-tests for her music theory students. A knowledge-gained score was...
-
Designing an Experiment Researchers wish to know if there is a link between hypertension (high blood pressure) and consumption of sugar. Past studies have indicated that the consumption of fruits...
-
Suppose there is a medical diagnostic test for a disease. The sensitivity of the test is .95 . This means that if a person has the disease, the probability that the test gives a positive response is...
-
Let \(Y\) have the Poisson \((=2)\) distribution. (a) Calculate \(P(Y=2)\). (b) Calculate \(P\left(\begin{array}{ll}Y & 2\end{array} ight)\). (c) Calculate \(P(1 \quad Y <4)\).
-
A pilot leaves an airfield heading due east and flies 130 miles. she then turns to face a different direction, at bearing N A W (for some angle A between 0 and 90 degrees). in this new direction, she...
-
On the basis of the details of the following fixed asset account, indicate the items to be reported on the statement of cashflows: ACCOUNT Land ACCOUNT NO. Balance Date Item Debit Credit Debit Credit...
-
Quantitatively compare the Carnot Rankine cycle of Figure 13.12(a) with the ideal Rankine cycle of Figure 13.12(b) Compare efficiencies and the work done per cycle. All the necessary information...
-
A sample of H 2 O at a pressure of 1 atm has a specific enthalpy of 700 kJ/kg. What is its temperature? What state is it in, a sub-cooled liquid, super heated vapor, or a mixed phase? If it is in a...
-
Can you identify the largest component of your personal electric power consumption?
-
In July 2017, Latrice Merritt entered a residential lease with Doran 610 Apartments, LLC. Under the terms of the lease agreement, Merritt was prohibited from installing a private security system in...
-
On March 13, 2009, Juan Mendez Sr. was admitted to a nursing facility. On that day, a doctor employed by the facility determined the father lacked the capacity to give informed consent or make...
-
In 2001, the City of New York and the Dormitory Authority of the State of New York (DASNY) entered into an agreement to build a forensic biology laboratory in Manhattan. Per the agreement, DASNY...

Study smarter with the SolutionInn App


