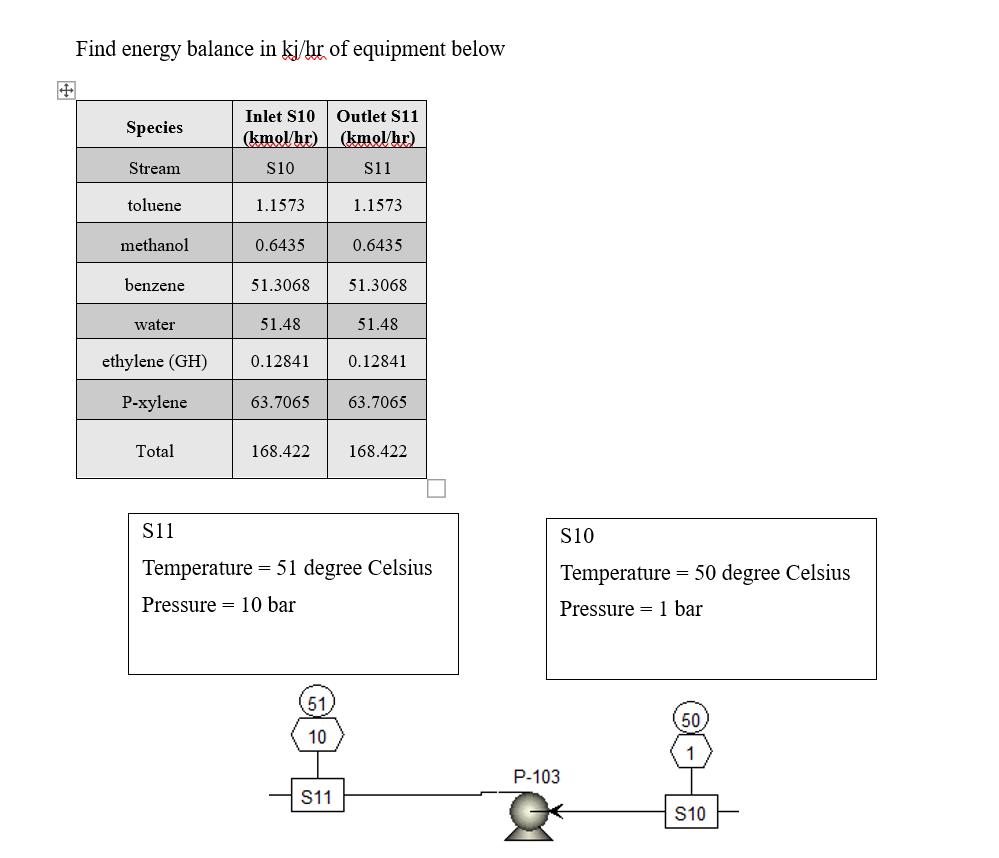
Find energy balance in kj/hr of equipment below Species Stream toluene methanol benzene water Total Inlet...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Find energy balance in kj/hr of equipment below Species Stream toluene methanol benzene water Total Inlet $10 Outlet S11 (kmol/hr) (kmol/hr) $10 $11 S11 1.1573 51.48 ethylene (GH) 0.12841 P-xylene 0.6435 51.3068 51.3068 63.7065 168.422 51 4-30 1.1573 10 0.6435 S11 51.48 0.12841 Temperature = 51 degree Celsius Pressure 10 bar 63.7065 168.422 S10 Temperature = 50 degree Celsius Pressure 1 bar P-103 50 1 S10 Find energy balance in kj/hr of equipment below Species Stream toluene methanol benzene water Total Inlet $10 Outlet S11 (kmol/hr) (kmol/hr) $10 $11 S11 1.1573 51.48 ethylene (GH) 0.12841 P-xylene 0.6435 51.3068 51.3068 63.7065 168.422 51 4-30 1.1573 10 0.6435 S11 51.48 0.12841 Temperature = 51 degree Celsius Pressure 10 bar 63.7065 168.422 S10 Temperature = 50 degree Celsius Pressure 1 bar P-103 50 1 S10
Expert Answer:
Answer rating: 100% (QA)
The reaction you provided involves the formation of an ester through a nucleophilic acyl substitutio... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A 2-metre long pipe is cut into 2 pieces. One piece is 2 times the length of the other piece. Find the length of each piece, entering your answers in metres and rounding your answers to two decimal...
-
The Horner-Emmons modification is a variation of a Wittig reaction in which a phosphonate-stabilized carbanion in used instead of a phosphonium ylide. The phosphonate-stabilized carbanion is prepared...
-
In the previous section, we learned how to use malonic ester as a starting material in the preparation of substituted carboxylic acids (the malonic ester synthesis). That method employed a step in...
-
Featherstone Inc. reported the following data: Net income ................................... $296,000 Depreciation expense ................... 113,100 Gain on disposal of equipment ...... 58,500...
-
On December 31, 2017, Dow Steel Corporation had 600,000 shares of common stock and 300,000 shares of 8%, noncumulative, nonconvertible preferred stock issued and outstanding. Dow issued a 4% common...
-
Listed below are the ages (years) of randomly selected race car drivers (based on data reported in USA Today). Use a 0.05 significance level to test the claim that the mean age of all race car...
-
You want to determine the mean age of all statistics instructors at the University of Colorado. representative Samples? In Exercises 1316, identify the sample, population, and sampling method. Then...
-
Mark Landman's accountant resigned and left the books in a mess. Mark is trying to compute unknown values in inventory accounts in three of his stores. Knowing of your expertise in cost flows, he...
-
Anna picks more oranges in an hour than Beth; Anna has the absolute advantage in oranges. Anna also picks more apples in an hour than Beth; Anna has the absolute advantage in apples. Comparative...
-
Is the bucket sort algorithm in place? why or why not public static void BSort(IntegerSequences) int n = $.size(): IntegerSequence Rosation prec, sarc for (int i=1; i succ.clement().intValue()) swap...
-
For the current model year, standards enacted under Trump require the fleet of new vehicles to get just under 28 miles per gallon in real-world driving. The new requirements increase gas mileage by...
-
Identify a true statement about heuristics: They require more scientific knowledge than algorithms. They are more complicated than algorithms. Unlike algorithms, they do not permit rapid solutions....
-
3. (a) What magnitude of magnetic field would make a beam of electrons, traveling to the right of this page at a speed of 4.8x106 m/s, go undeflected through a region where there is a uniform...
-
Write a few sentences about C# or Project Management. Did you learn anything new when researching these topics? Did you find a helpful site or article that you can share with class? Second Post:...
-
Conduct stock valuations for Tesla using a set of techniques such as the Single-Index market model and Discounted Dividend Model (DDM). Single-Index Market Model: The Single-Index market model is a...
-
On 1/1/2019, the city of San Francisco issued at par $2,000,000 of 4% term bonds to renovate the Golden Gate Bridge. The bonds mature in five years on 1/1/2024 with semiannual interest payments on...
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
Using the pKa values in Table 17.1, predict the products of the following reactions: a. b. c. d. OCH3 CH3 NaOH CH3 Table 17.1 The pKa Values of the Conjugate Adds of the Leaving Groups of Carbonyl...
-
What products would be obtained from the reaction of 1,3,5-hexatriene with one equivalent of HBr? Ignore stereoisomers.
-
Which of each pair of diols is cleaved more rapidly by periodic acid? a. b. OH OH or OH DHH C(CH3)3 or C(CH3)3 CH3)3
-
The financial statements for the business of Jets Ski Equipment are shown below. Additional information 1. All purchases and sales of inventories are on credit. 2. On 1 July 2019, J. Waters injected...
-
Some of the most recent financial statements for Hyland Pty Ltd are shown below. Additional information 1. All purchases and sales of inventories are on credit. 2. On 1 July 2020, the shareholders...
-
The comparative statements of financial position of Cresta Ltd as at 30 June 2019 and 2020, and the income statement and statement of changes in equity for the year ended 30 June 2020 are shown...

Study smarter with the SolutionInn App


