In Intrinsic Ste GaAs e and hele mobilities are. 0.85 & 0.04 met / Usec...
Fantastic news! We've Found the answer you've been seeking!
Question:
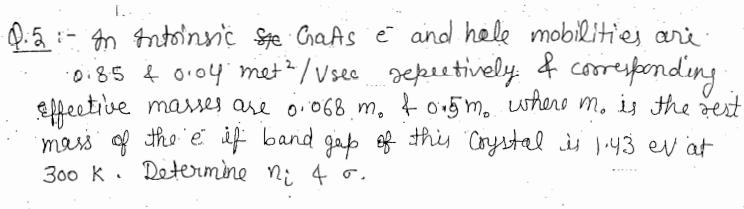
Transcribed Image Text:
In Intrinsic Ste GaAs e and hele mobilities are. ·0.85 & 0.04 met ² / Usec repectively. & corresponding. effective masses are 0.068 m. &0.5m. where m. is the rest mass of the e if band gap of this Crystal is 1.43 ev at 300 K. Determine ni 4 o. L In Intrinsic Ste GaAs e and hele mobilities are. ·0.85 & 0.04 met ² / Usec repectively. & corresponding. effective masses are 0.068 m. &0.5m. where m. is the rest mass of the e if band gap of this Crystal is 1.43 ev at 300 K. Determine ni 4 o. L
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
Calculation the rest mass of a proton in MeV/c2
-
A proton has 1836 times the rest mass of an electron. At what speed will an electron have the same momentum as a proton moving at 0.0100c?
-
According to Table 12.4, the rest mass of an electron is 0.511 MeV/c 2 . What is the rest mass of a positron? Table 12.4 Mass (MeV/c) Symbol Family Electron Antiparticle e+ (positron) Particle Name...
-
Question 4: What is meant by statistical versus nonstatistical and probabilistic versus nonprobabilistic sample selection?
-
a. If Fama Company, with a break-even point at $360,000 of sales, has actual sales of $480,000, what is the margin of safety expressed (1) in dollars and (2) as a percentage of sales? b. If the...
-
Aldovar Company produces a variety of chemicals. One division makes reagents for laboratories. The division's projected income statement for the coming year is: Sales (203,000 units @ $70)...
-
Share capital is: a. The way in which capital is divided at the end of each year b. The way in which partners decide to divide profits and losses c. Another name for London's stocl~ exchange d. The...
-
Boston's famous Limoges Restaurant is open 24 hours a day. Servers report for duty at 3 A.M., 7 A.M., 11 A.M, 3 P.M., 7 P.M., or 11 P.M., and each works an 8-hour shift. The following table shows the...
-
Canada Ltd was incorporated on 1 January 2018 and on that date issued: 5% Preference shares for $10 4 000 shares Ordinary shares for $100 5 000 shares During December 2020 Canada Ltd declared a total...
-
Juliana Oliveria needs additional filing space at the end of the year in the companys office and chooses to use off-site, secured storage. Upon arriving at the storage facility, she discovers that...
-
. Question 1 1 pts 1. What was Cindy's Finest margin as a percentage for years 3, 4, and 5? O 69%, 9.65%, 11.15% 0 92%, 12.87%, 14.86% 0 4.35%, 10.77%, 13.56% 0 43.48%, 46.15%, 45.76% Question 2 1...
-
Haminder has just been hired at a new job and is planning to pay off his credit card debt of $20,000. He plans to make equal monthly payments for 48 months, starting next month. His bank has quoted...
-
Chevelle, Inc., has sales of , costs of , depreciation expense of , and interest expense of . If the tax rate is 21 percent, what is the operating cash flow, or OCF? Sales 39,500 Costs 18,400...
-
Syntec Company uses a job-order costing system and applies overhead based on direct labor dollars. Job M45 was started in March and incurred $1,950 in direct material cost, $1,780 in direct labor...
-
a. The largest single-day point loss of the Dow jones Industrial Average occurred on September 29, 2008, when the market lost 778 points and closed at 10,365. What was the percentage change? b. The...
-
You purchase $100,000 of par value of a TIPS with a real rate of 4.5%. Over the first 6 months after issuance the annualized inflation rate is 8.2% and over the following six months it is 8.5%. What...
-
Assume that the $500,000 mortgage on Wheeler's real property is nonrecourse, rather than recourse, so that the mortgage holder legally may look oniy to the building, plant, and land for satisfaction...
-
During registration at Tech every quarter, students in the Department of Management must have their courses approved by the departmental advisor. It takes the advisor an average of 4 minutes...
-
Find the fraction of ionized donor impurities if the donor ionization energy is large enough that d is larger than log(nc/8nd) by several times . The result explains why substances with large...
-
(a) Use the partition function to find an exact expression for the magnetization M and the susceptibility X = dM/dB as a function of temperature and magnetic field for the model system of magnetic nm...
-
Consider a plane sheet of material or absorptivity a, emissivity e, and reflectivity r = 1- a. Let the sheet be suspended between and parallel with two black sheets maintained at temperatures u and...
-
Griffin Metals Co. has provided the following data: The selling price is expected to be \($300\) per tonne for the first six months and \($310\) per tonne thereafter. Variable costs per tonne are...
-
Highjinks Corporation's sales department has estimated revenue of 250,000 for your division. 60% of this will be achieved in the first half-year and 40% in the remaining half-year. Variable operating...
-
The total cost of goods sold for June is a. $103,500 b. $128,800 c. 57,500 d. $232,300

Study smarter with the SolutionInn App



