In the industrial synthesis of hydrogen, mixtures of CO and H2 are enriched in H2 by...
Fantastic news! We've Found the answer you've been seeking!
Question:
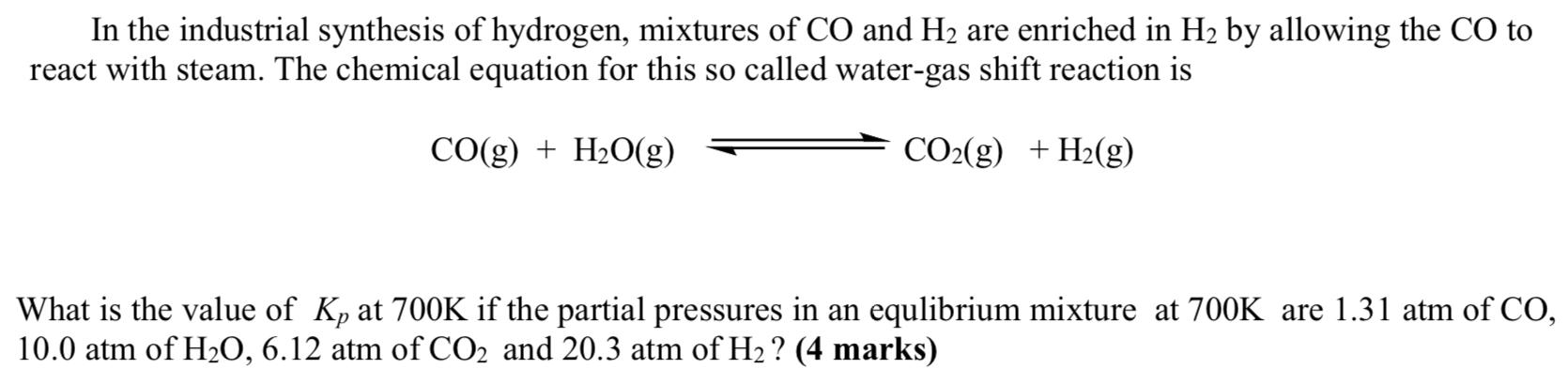
Transcribed Image Text:
In the industrial synthesis of hydrogen, mixtures of CO and H2 are enriched in H2 by allowing the CO to react with steam. The chemical equation for this so called water-gas shift reaction is CO(g) + H2O(g) CO2(g) +H2(g) What is the value of Kp at 700K if the partial pressures in an equlibrium mixture at 700K are 1.31 atm of CO, 10.0 atm of H2O, 6.12 atm of CO2 and 20.3 atm of H2 ? (4 marks) In the industrial synthesis of hydrogen, mixtures of CO and H2 are enriched in H2 by allowing the CO to react with steam. The chemical equation for this so called water-gas shift reaction is CO(g) + H2O(g) CO2(g) +H2(g) What is the value of Kp at 700K if the partial pressures in an equlibrium mixture at 700K are 1.31 atm of CO, 10.0 atm of H2O, 6.12 atm of CO2 and 20.3 atm of H2 ? (4 marks)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The industrial synthesis of methyl tert-butyl ether involves treatment of 2-methylpropene with methanol (CH3OH) in the presence of an acid catalyst, as shown in the following equation. CH3 H3C H3C...
-
Benzene reacts with bromine. a. Write a balanced chemical equation for this reaction. b. Name the catalyst used. c. What visual observations could be made during the reaction? d. Benzene will also...
-
For the chemical reaction CO2 + H2 CO + H2O, the equilibrium value of the degree of reaction (forward completion fraction) at 1200 K is 0.56. Determine the equilibrium constant and the change in...
-
The rigid bars ABC and CD are supported by pins at A and D and by a steel rod at B. There is a roller connection between the bars at C. Compute the vertical displacement of point C caused by the...
-
An online investment blogger advises investing in mutual funds that have performed badly the past year because regression to the mean tells us that they will do well next year. Is he correct?
-
Whole Foods strives to be an Ethical Corporate Citizen. Write 1-2 page about your opinion either agreeing or disagreeing with the article, and why it is important to business.
-
Define anti-patterns, functional decomposition and the relationship between them.
-
1. Banks are required by law to keep a fraction of their deposits as ______. 2. When reserves do not pay interest, banks prefer to keep reserves rather than make loans. ______ (True/False) 3. If the...
-
If there were a shift of $8,000 in sales revenue from the banquet area to the dining room, would you expect the restaurant's overall operating income to increase or decrease? Explain your reasoning...
-
As the in-charge senior auditor on the audit engagement for JA Tire Manufacturing for the year ended December 31, 2019, you are responsible for performing risk assessment procedures related to the...
-
A thin uniform rod is initially positioned in the vertical direction, with its lower end attached to a frictionless axis that is mounted on the floor. The rod has a length of 1.70m and is allowed to...
-
Sometimes, lenders allow or require a downpayment before they extend you the loan. What would be the advantage to the lender? What would be the advantage to the borrower?!
-
The units of an item available for sale during the year were as follows: 11 units @ $34 Jan. 1 Inventory Feb. 17 Purchase 12 units @ $36 July 21 Purchase Nov. 23 Purchase 4 units @ $38 17 units @ $39...
-
A company has 410,000 basic shares outstanding and a current stock price of $4000. A financial buyer decides to offer a 65% premium to the current share price. Calculate the equity purchase price in...
-
Daphne bought a house ten years ago for $245,000.The real estate market has had it ups and downs since then. Daphne read a story in todays newspaper that says that during the past decade, houses in...
-
From a friend who is Human Resources Manager for a law fi rm: I need your help. Our partners are contemplating the introduction of 360-degree appraisal, and have asked my advice about the benefi ts...
-
Use Warshall's algorithm to compute the transitive closure of the graph with the following adjacency matrix: 0 |1 0 0 [Select ] 0 |1 0 Choose the correct values for the unknown variables after each...
-
If a test has high reliability. O the test measures what the authors of the test claim it measures O people who take the same test twice get approximately the same scores both times O scores on the...
-
Explain how you could distinguish between 1-hexyne and 4-methyl-2-pentyne by 13C NMR.
-
Indole (Thble 25.1) in many cases undergoes electrophilic aromatic substitution at carbon-3. Using this observation, give the structure of the azo dye formed in the following reaction. NaNO, H S04...
-
What rate law would be expected for the reaction of cyanide ion -:CN) with ethyl bromide b: the SN2 mechanism?
-
What is the role of the origin of transfer during conjugation involving F+ and Hfr strains? What is the significance of the direction of transfer in Hfr conjugation?
-
What is the role of sex pili during conjugation?
-
Think about the structure and transmission of F factors, and discuss how you think F factors may have originated.

Study smarter with the SolutionInn App



