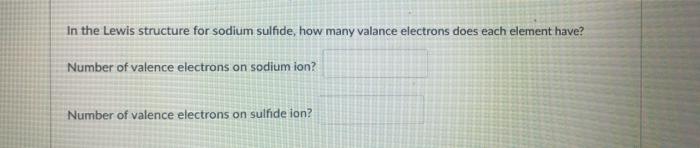
In the Lewis structure for sodium sulfide, how many valance electrons does each element have? Number...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In the Lewis structure for sodium sulfide, how many valance electrons does each element have? Number of valence electrons on sodium ion? Number of valence electrons on sulfide ion? D Question 55 Acids always have hydrogen(s) at the I of the chemical formula. 2 pts In the Lewis structure for sodium sulfide, how many valance electrons does each element have? Number of valence electrons on sodium ion? Number of valence electrons on sulfide ion? D Question 55 Acids always have hydrogen(s) at the I of the chemical formula. 2 pts
Expert Answer:
Answer rating: 100% (QA)
Number of valence electrons on sodium ion 8 electrons Number of valence electrons on sulfide ion 8 ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
In the Lewis structure shown here, A, D, E, Q, X, and Z represent elements in the first two rows of the periodic table (H--- Ne). Identify all six elements so that the formal charges of all atoms are...
-
The Lewis structure for allene is Make a sketch of the structure of this molecule that is analogous to Figure 9.25. In addition, answer the following three questions: (a) Is the molecule planar? (b)...
-
Consider the Lewis structure for the polyatomic oxyanion shown here, where X is an element from the third period (Na --- Ar). By changing the overall charge, n, from 1 -- to 2 --- to 3 --- we get...
-
In the figure below, a square of edge lengths is formed by four spheres of masses, m, M, m3, and m4. What is the x component and the y component of the net gravitational force from them on a central...
-
Northern Refineries does not avoid risk by selling oil futures. If prices stay above $1.60 a gallon, then it will actually have lost by selling oil futures at that price. Is this a fair comment?
-
LAW: It is said that only parties to a contract are compelled to observe the contractual abligation , howover . this is not entirely true simply because every general nule has an exception ....
-
Widget, Corp., completed the following inventory transactions during the month of January: Requirements 1. Without resorting to calculations, determine which inventory method will result in Widget,...
-
The following information describes transactions for Morgenstern Advertising Company during July: a. On July 5, Morgenstern purchased and received $21,500 of supplies on credit from Drexel Supply...
-
Discuss about the safety awareness program implemented by your organization in order to meet the requirement of Occupational Health and Safety Administration.
-
Usonic, Inc., has collected the following information on its cost of electricity: Required a. Prepare a scattergraph of Usonic's electricity costs for the year. Plot the total electricity cost on the...
-
Below are the first several terms of three different sequences. For each one identify if the sequence is arithmetic, geometric or neither. Also for each sequence identify the nth term in the...
-
Let U (a, b, c, d, e, f, g, h, k, m}, A = {g, k, m), B = {b, k, m), and C = {b, d, e, f, h}. Find the following set. AU (BNC)
-
1. A company purchased machinery in 2015 for $400,000. Its value in 2018 was $320,000. Assuming the resale value decreases exponentially, what will the value be in 2020? As a part of your solution,...
-
You put $1,000 into a bank account today and then add $100 at the end of each of the next 5 years, with a 7% interest rate, how much will you have on the date of the last payment?
-
The fact that Apple has no manufacturing facilities of its own Question 21 options: has been problematic for Apple in terms of debt. has caused it to build up massive debt on its balance sheet. has...
-
More recently, studies have found evidence suggesting that many executives have engaged in a more direct form of manipulating their stock option compensation: backdating their option grants. What is...
-
Convert the following to decimal. a. b. 1110 0100 (8-bit two's complement) 4A CB16 (unsigned) C. 5098 (9-bit two's complement) d. 31325 (9-bit two's complement) e. 1101 1111 1001 (12-bit two's...
-
Find the inverse, if it exists, for the matrix. -1
-
Natural gas consists primarily of methane, CH4(g). (a) Write a balanced chemical equation for the complete combustion of methane to produce CO2(g) as the only carbon-containing product. (b) Write a...
-
Germanium has the same structure as silicon, but the unit cell size is different because Ge and Si atoms are not the same size. If you were to repeat the experiment described in the previous problem...
-
Fill in the gaps in the following table: 59 Co3+ Symbol Protons Neutron:s Electrons Net charge 34 46 36 76 80 120 78 116 2+
-
The Excel formula to compute net income's percentage of sales in Exhibit 7-5, is a. \(=B 4 * B 2\) b. \(=B 4 / B 2\) c. \(=\mathrm{B} 2-\mathrm{B} 3\) d. \(=\mathrm{B} 4+\mathrm{B} 3\) EXHIBIT 7-5 A...
-
Match each component of a computerized accounting system with its meaning. Meaning Component Electronic linkages that allow different computers A. Server to share the same information Electronic...
-
Complete the crossword puzzle that follows. 2 3 4 5 6 Down: 1. Managers need over operations in order to authorize transactions and safeguard assets 3. Programs that drive a computer 4. Electronic...

Study smarter with the SolutionInn App


