Question: = In the phase diagram for water, indicate the direction that the solid-liquid and liquid-gas coexistence lines will move along the temperature axis after
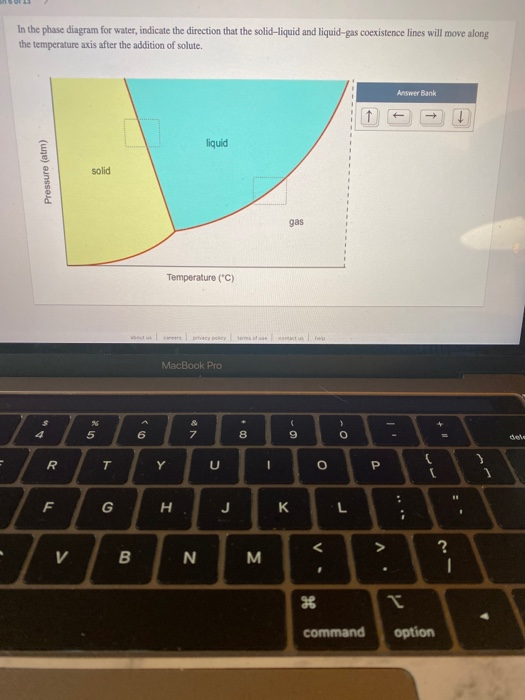
= In the phase diagram for water, indicate the direction that the solid-liquid and liquid-gas coexistence lines will move along the temperature axis after the addition of solute. Pressure (atm) R F V solid % 5 T G B 6 Temperature (C) careers privacy policy ma MacBook Pro Y H liquid & 7 N N U J 00+ 8 M I gas contact us help ( - 9 K O V. < H > O L command P ^. Answer Bank : * " I ( option +11 ? 11 I dele
Step by Step Solution
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Step 1 When a nonvolatile solute is added to a solvent t here will be an elevation i... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


