In the presence of an acid catalyst, cyclohexanol (100.16 g/mol) is dehydrated to give cyclohexane (82.14...
Fantastic news! We've Found the answer you've been seeking!
Question:
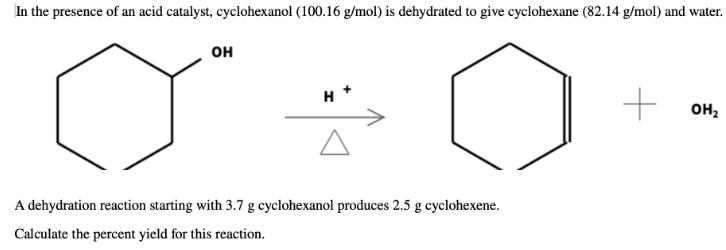
Transcribed Image Text:
In the presence of an acid catalyst, cyclohexanol (100.16 g/mol) is dehydrated to give cyclohexane (82.14 g/mol) and water. он н он, A dehydration reaction starting with 3.7 g cyclohexanol produces 2.5 g cyclohexene. Calculate the percent yield for this reaction. In the presence of an acid catalyst, cyclohexanol (100.16 g/mol) is dehydrated to give cyclohexane (82.14 g/mol) and water. он н он, A dehydration reaction starting with 3.7 g cyclohexanol produces 2.5 g cyclohexene. Calculate the percent yield for this reaction.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A carbohydrate (S) decomposes in the presence of an enzyme (E) The Michaelis-Menten kinetic parameters were found to be as follows M 200 mol m 3 100 mol m 3 min a Calculate the change of substrate...
-
On heating 1,2,4-butanetriol in the presence of an acid catalyst, a cyclic ether of molecular formula C4H8O2 was obtained in 81-88% yield. Suggest a reasonable structure for this product.
-
In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals in large doses, paraldehyde is used as a sedative or...
-
Which description does NOT fit in description of "issues" in the context of international standards? a. An unsettled matter. b. A vital matter. c. A change in the environment. d. A concern or...
-
Draw the general structure of a synovial joint labeling all the mail parts.
-
An aggressive financial planner who claims to have a superior method for picking undervalued stocks is courting one of your clients. The planner claims that the best way to find the value of a stock...
-
Refer to the information in Problem 21-1B. Tohono Companys actual income statement for 2017 follows. Required 1. Prepare a flexible budget performance report for 2017. Analysis Component 2. Analyze...
-
Financial Statements Circus City issued an 8%, 10-year $2,000,000 bond to build a monorail mass transit system. The city received $1,754,217 cash from the bond issuance on January 1, 2012. The bond...
-
The index of refraction for a particular material is 1.38. Determine the speed of light in this medium. Light passes from water (n=1.33) into air (n=1.003) at an angle of incidence of 25 degrees....
-
The current assets and liabilities sections of the comparative balance sheets of Regent Inc., a private entity reporting under ASPE, at December 31 are presented below, along with the income...
-
If you have a gross annual salary of $75,000 and a net annual salary of $50,000, what is the maximum amount that you can spend on your mortgage payment (principal and interest), your property taxes...
-
What is the difference between PPE as per Ind AS 16 and owner occupied property as defined in Ind AS 16?
-
Fairfield Companys payroll costs for the most recent month are summarized here: Item Description Total Cost Hourly labor wages 850 hours @ $27 per hour 170 hours for Job 101 = $ 4,590 330 hours for...
-
Hilltop School District receives a donation of ten computers from Computer Hut, a local computer firm.. The cost of Computer Hut of each computer is $2,500. The retail value of each computer is...
-
I. True/False/Uncertain - Briefly explain. No credit without an explanation (8 marks each). 1. At the 10th decile the (cumulative) lift would never be one. 2. With 4 categories of Y and 5 Xs, the...
-
A block of mass m = 5.4 kg slides on a rough surface and moves toward a spring with a spring constant k, as shown in the figure below. When the block is d = 13.6m away from the spring, it has a...
-
Do corporate officers and directors have a duty to manage the corporation for the purpose of maximising profits for the benefit of shareholders or stakeholders? Discuss with reference to the US case...
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
Suggest two ways in which each of the following alcohols might be prepared by using a Grignard reagent: 2-Phenyl-2-propanol, CGHs(CH3)2
-
Give the structure of the product formed when each of the following alkenes reacts with bromine in water: (a) 2-Methyl-2-butene (b) 3-Methyl-1-butene (c) 1-Methylcyclopentene
-
A sample of synthetic cholesterol was prepared consisting entirely of the enantiomer of natural cholesterol. A mixture of natural and synthetic cholesterol has a specific rotation []D20 of -13. What...
-
Verify the color SU(3) representations for combinations of three or fewer quarks and antiquarks given in Eq. (19.28). Data from Eq. 19.28 qq=303=108 qq 3 3 603, 999 3 3 3 = 36315, qqq 3 3 3 1088 10,
-
Prove that Eq. (19.34) gives the simplest multi-gluon and gluon-quark states that contain an \(\mathrm{SU}(3)\) color singlet in the decomposition. Data from Eq. 19.34 (GG)1: (88)1 (Gqq) : [8 (383)8]...
-
A textile manufacturer is concerned about the strength of cotton yarn. In order to nd out whether fiber length is an important factor in determining the strength of yarn, the quality control manager...

Study smarter with the SolutionInn App



