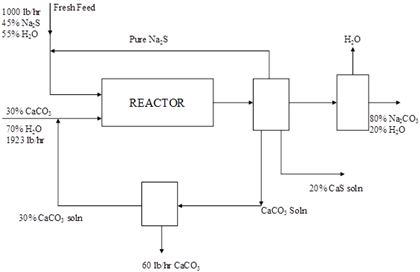
In the process sketched in Figure, Na2CO3 is produced by the reaction Na2S + CaCO3 ...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
In the process sketched in Figure, Na2CO3 is produced by the reaction Na2S + CaCO3 → Na2CO3 + CaS. The reaction is 90% complete on one pass through the reactor, and the amount of CaCO3 entering the reactor is 50% in excess of that needed. Calculate on the basis of 1000 lb/hr of fresh feed (a) the pounds of Na2S recycled, and (b) the pounds of Na2CO3 solution formed per hour. 1000 lb/hr 45% Na S 55% H₂O 30% CaCO₂ 70% H₂O 1923 lb hr Fresh Feed 30% CaCO, soln Pure Na $ REACTOR 60 lb/hr CaCO, CaCO, Soln H₂O 20% CaS soln $0% NaCO₂ 20% H₂O In the process sketched in Figure, Na2CO3 is produced by the reaction Na2S + CaCO3 → Na2CO3 + CaS. The reaction is 90% complete on one pass through the reactor, and the amount of CaCO3 entering the reactor is 50% in excess of that needed. Calculate on the basis of 1000 lb/hr of fresh feed (a) the pounds of Na2S recycled, and (b) the pounds of Na2CO3 solution formed per hour. 1000 lb/hr 45% Na S 55% H₂O 30% CaCO₂ 70% H₂O 1923 lb hr Fresh Feed 30% CaCO, soln Pure Na $ REACTOR 60 lb/hr CaCO, CaCO, Soln H₂O 20% CaS soln $0% NaCO₂ 20% H₂O
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Chlorine dioxide (ClO 2 ), which is produced by the reaction 2NaClO 2 (aq) + Cl 2 (g) 2ClO 2 (g) + 2NaCl(aq) has been tested as a disinfectant for municipal water treatment. a. Using data from Table...
-
Placid Places Incorporated, a calendar year taxpayer, purchased an apartment building on October 1 of the current year for $1,200,000, of which $200,000 was allocable to the land. The corporation's...
-
Figure a shows a wire that forms a rectangle (W = 20 cm, H = 30 cm) and has a resistance of 5.0m??). Its interior is split into three equal areas, with magnetic fields B1, B2, and B3. The fields are...
-
Using the SEDAR database, find the most recent annual reports for two Canadian retailers (e.g., Loblaw, Rona, Danier Leather). Required: a. Based on the information provided in the companies audited...
-
It has been stated that it is generally undesirable to allow temperatures to rise excessively in machining operations. Explain why.
-
Do you think people and organizations can ever become gender- and color-blind? Discuss.
-
Petitioner Salman was indicted for federal securities-fraud crimes for trading on inside information he received from a friend and relative-by-marriage, Michael Kara, who, in turn, had received the...
-
(EPS: Simple Capital Structure) On January 1, 2010, Bailey Industries had stock outstanding as follows. 6% Cumulative preferred stock, $100 par value, issued and outstanding 10,000 shares $1,000,000...
-
You are to PAY 350,000 on March 16. Todays spot rate is $1.15/. The forward rate is $1.18/. On March 16 the spot rate is $1.14/. Futures contracts are for 125,000 each. Q1. How many contracts do you...
-
Using the Internet, find two examples of advertisements for sports products that use indirect objectives and two examples of advertisements that use direct objectives.
-
Write a critical review on the following video either from the perspective of Existentialism or Pragmatism. Your review should not exceed more than 100 words. https://youtu.be/765joQGfglA
-
Finance House ( FH ) sold an equipment, costing Rs . 6 0 , 0 0 0 , to a customer on installment sale basis on January 1 , 2 0 1 8 . Each installment of Rs . 4 0 , 0 0 0 will be received on every...
-
Disclaimer for any procedure-driven, calculation-involved questions below, please show problem-solving procedure to earn full credit. A human resource manager of a large company wants to determine...
-
Choose a case of financial fraud or financial misconduct that has happened lately. Identify examples of the three ingredients of the fraud triangle that were in place that allowed the fraud to take...
-
Flounder Corporation began operating as a business in 2 0 2 0 . During January 2 0 2 1 , the company paid $ 2 9 6 5 0 0 in design costs to develop its trademark and $ 2 4 4 0 0 0 in legal and...
-
On 2 / 2 , 2 / 2 , maintenance services were provided to the Rabwa facility for an amount of 1 6 0 , 0 0 0 , 1 6 0 , 0 0 0 , which was received by check.It is required to make daily entries and then...
-
I have an assignment called an Detection Investigative Project Design where, can you please go through and answered them accordingly as a paragraph? Investigative Project Design: Part 1 This aspect...
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
The unit cell of MgO is shown below. Does MgO have a structure like that of NaCl or ZnS? If the density of MgO is 3.58 g/cm3, estimate the radius (in centimeters) of the O2- anions and the Mg2+...
-
Ozone is a possible replacement for chlorine in municipal water purification. Unlike chlorine, virtually no ozone remains after treatment. This has good and bad consequences. Explain.
-
The reaction 2NO(g) + Br2(g) 2NOBr(g) as Kp = 109 at 25oC. If the equilibrium partial pressure of Br2 is 0.0159 atm and the equilibrium partial pressure of NOBr is 0.0768 atm, calculate the partial...
-
Discuss the importance of validity and research design. Next, choose one type of validity (internal, external, construct, or statistical conclusion) and discuss its relevance to experimental, quasi...
-
Explain from a technical viewpoint why it is important to distinguish a method, research, approach, and design. Next, briefly discuss how understanding each term individually in addition to how these...
-
Define the two major types of sampling strategies .

Study smarter with the SolutionInn App


