Question: Indicate the relationship between the two structures in the pair. Are they chair conformations of the same molecule? If so, are they conformational diastereomers,
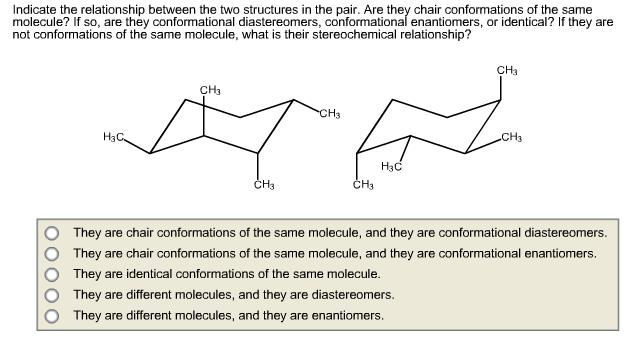
Indicate the relationship between the two structures in the pair. Are they chair conformations of the same molecule? If so, are they conformational diastereomers, conformational enantiomers, or identical? If they are not conformations of the same molecule, what is their stereochemical relationship? CHa CH3 CH3 H3C CH3 H3C CH3 They are chair conformations of the same molecule, and they are conformational diastereomers. They are chair conformations of the same molecule, and they are conformational enantiomers, They are identical conformations of the same molecule. They are different molecules, and they are diastereomers. They are different molecules, and they are enantiomers. O000
Step by Step Solution
There are 3 Steps involved in it
It seems that no images are upload... View full answer

Get step-by-step solutions from verified subject matter experts


