Question: Indicate which ion is preferred by soil in each graph. 1.0 0.8- 0.6-- 0.4 K-Na I = 0.010 0.00 0.0 0.2 0.4 0.6 0.8 1.0
Indicate which ion is preferred by soil in each graph.
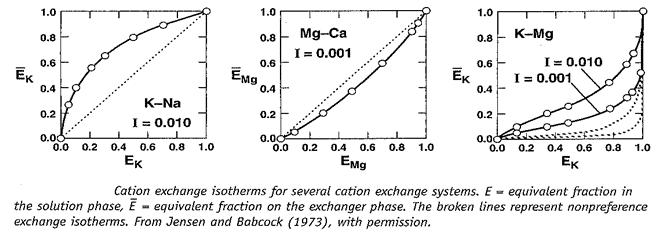
1.0 0.8- 0.6-- 0.4 K-Na I = 0.010 0.00 0.0 0.2 0.4 0.6 0.8 1.0 0.2 1.0 0.8- 0.6- 0.4 EMg 0.2 Mg-Ca I = 0.001 0.00 0.0 0.2 0.4 0.6 0.8 1.0 EMg k 1.0 0.8 0.6- 0.4-- K-Mg I= 0.010 I = 0.001 0.2 0.00 0.0 0.2 0.4 0.6 0.8 1.0 Cation exchange isotherms for several cation exchange systems. E equivalent fraction in the solution phase, equivalent fraction on the exchanger phase. The broken lines represent nonpreference exchange isotherms. From Jensen and Babcock (1973), with permission.
Step by Step Solution
3.52 Rating (155 Votes )
There are 3 Steps involved in it
The graphs youve provided show cation exchange isotherms for different ion exchange systems between pairs of ions KNa MgCa and KMg Here E represents t... View full answer

Get step-by-step solutions from verified subject matter experts


