Instructions: You are to place the items below in their proper place in the statement of...
Fantastic news! We've Found the answer you've been seeking!
Question:
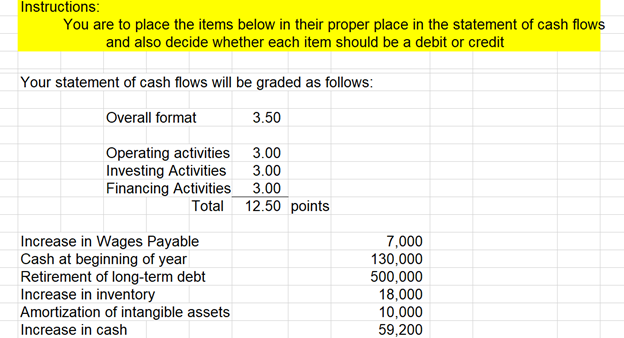


Transcribed Image Text:
Instructions: You are to place the items below in their proper place in the statement of cash flows and also decide whether each item should be a debit or credit Your statement of cash flows will be graded as follows: Overall format 3.50 Operating activities 3.00 Investing Activities 3.00 Financing Activities 3.00 Total 12.50 points Increase in Wages Payable 7,000 Cash at beginning of year 130,000 Retirement of long-term debt 500,000 Increase in inventory 18,000 Amortization of intangible assets 10,000 Increase in cash 59,200 Purchase machinery 62,000 Increase in accounts receivable 19,000 increase in interest payable 5,900 Bonds exchanged for Common Stock 45,000 Dividends paid 22,000 Decrease in accounts payable 23,400 Net income 450,700 Sale of common stock 160,000 Depreciation expense 70,000 The Staples Corporation Statement of Cash Flows As of December 31, 2023 Indirect Method Instructions: You are to place the items below in their proper place in the statement of cash flows and also decide whether each item should be a debit or credit Your statement of cash flows will be graded as follows: Overall format 3.50 Operating activities 3.00 Investing Activities 3.00 Financing Activities 3.00 Total 12.50 points Increase in Wages Payable 7,000 Cash at beginning of year 130,000 Retirement of long-term debt 500,000 Increase in inventory 18,000 Amortization of intangible assets 10,000 Increase in cash 59,200 Purchase machinery 62,000 Increase in accounts receivable 19,000 increase in interest payable 5,900 Bonds exchanged for Common Stock 45,000 Dividends paid 22,000 Decrease in accounts payable 23,400 Net income 450,700 Sale of common stock 160,000 Depreciation expense 70,000 The Staples Corporation Statement of Cash Flows As of December 31, 2023 Indirect Method
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
MTI, a global telecommunications company, manufactures cable boxes and DVRs for its customers. Demand varies considerably from quarter to quarter. Using the demand, capacities, and cost figures given...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
In Problem 3.67 on page 135, you were introduced to a tea-bag-filling operation. An important quality characteristic of interest for this process is the weight of the tea in the individual bags. The...
-
1. Explain the role that marketing research played in the creation and launch of Domino's new pizza. 2. Are there more effective ways that Domino's could have gone about its research process? 3. Why...
-
Which of the following provides an example of divisions based on a functional organizational structure? a. Americas, Africa, Asia, Europe b. Research and development, production, finance, marketing...
-
T. Christian Cooper was a partner to Sanders and Richard Campbell d/b/a The Mullen Company. In 2001, Cooper helped bring about a management agreement between The Mullen Co. and Newnan Crossing...
-
In the United States, accounting for pensions has received a great deal of attention. In other countries, pension accounting is given much less attention. In one page, examine the reasons that would...
-
Complete a detailed Income Statement using the information from the Trial Balance. Make sure to list out all accounts you use to complete the Income Statement. TRIAL BALANCE AS OF DECEMBER 31, 20XX...
-
Study the following UNIX filesystem tree carefully and answer questions a- d: bin myfirst report lib david usr daniel dev gabriel a. Given that your present working directory (pwd) is usr, write down...
-
Draw a logic circuit diagram of a 4-bit 2S complement full adder or subtractor and hence describe its operation
-
What is the present value of $ 1 5 , 5 0 0 per year ( end of the year ) for 1 0 years assuming a discount rate of 7 . 5 % compounded annually?
-
A isolated conducting sphere of radius 9cm, initially uncharged, is illuminated by ultraviolet light of wavelength 280nm. What charge will the photoelectric effect induce on the sphere? The work...
-
Your firm is considering investing $ 7 5 0 , 0 0 0 into a project. This project will then generate cash flows of $ 1 2 8 , 0 0 0 , $ 1 5 6 , 0 0 0 , $ 2 4 8 , 0 0 0 , $ 1 7 4 , 0 0 0 , $ 2 6 0 , 0 0...
-
a) Write a simple timetabling program that asks the user to type in the number of students that are expected to be in class not conting the lecturer, assuming that the number typed in is n, display...
-
You will identify the neuromuscular junction parts indicate where botulism toxin acts. Part A Label the neuromuscular junction and indicate where the botulism toxin acts. Drag the appropriate labels...
-
ABC company leased new advanced computer equipment to STU Ltd on 1 January 2019.STULtd has to pay annual rental of $290,000 starting at 1 January 2019. It is a four years lease with ultimate rental...
-
Return to Targets 201 0 annual report. For instructions on how to access the report online, see the Continuing Financial Statement Analysis Problem in Chapter 2. On page 33 of the annual report youll...
-
This exercise continues the accounting for Lydons Yard Care, Inc., from the Continuing Exercise in Chapter 10. Assume that Lydons Yard Care, Inc., had the following comparative balance sheet at the...
-
Purpose: To help you understand the importance of cash flows in the operation of a small business. Its the end of the month and cash flow has been a little slow, as it usually is during this time of...
-
In a steam power plant, coal is burned at the rate of 50 lbm/min. The percentage of ash in the coal is 9% by mass. The combustion of coal takes place in a fluidized bed reactor and the ash in the...
-
2-Mercaptoethanol (C 2 H 6 OS) is produced by reacting ethylene oxide (C 2 H 4 O) with hydrogen sulfide (H 2 S) as per the following reaction equation. The reaction takes place in the presence of...
-
Chlorobenzene is produced by direct chlorination of benzene. However, a parallel reaction that produces dichlorobenzene hexachloride also occurs. The parallel reactions are shown here. The single...

Study smarter with the SolutionInn App


