Internal standards are commonly used in chromatography to calculate the concentration of an analyte. For any...
Fantastic news! We've Found the answer you've been seeking!
Question:
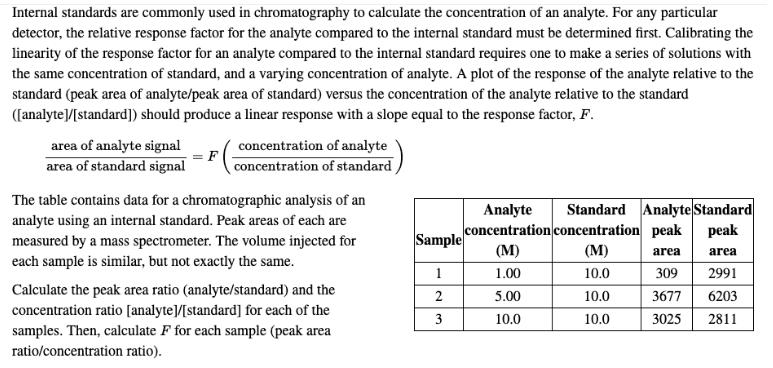
Transcribed Image Text:
Internal standards are commonly used in chromatography to calculate the concentration of an analyte. For any particular detector, the relative response factor for the analyte compared to the internal standard must be determined first. Calibrating the linearity of the response factor for an analyte compared to the internal standard requires one to make a series of solutions with the same concentration of standard, and a varying concentration of analyte. A plot of the response of the analyte relative to the standard (peak area of analyte/peak area of standard) versus the concentration of the analyte relative to the standard ([analyte]/[standard]) should produce a linear response with a slope equal to the response factor, F. area of analyte signal concentration of analyte area of standard signal concentration of standard The table contains data for a chromatographic analysis of an analyte using an internal standard. Peak areas of each are measured by a mass spectrometer. The volume injected for Standard Analyte Standard Analyte concentration concentration peak (M) pea Sample (M) area area each sample is similar, but not exactly the same. 1.00 10.0 309 2991 Calculate the peak area ratio (analyte/standard) and the concentration ratio [analyte]/[standard] for each of the samples. Then, calculate F for each sample (peak area ratio/concentration ratio). 5.00 10.0 3677 6203 3 10.0 10.0 3025 2811 Internal standards are commonly used in chromatography to calculate the concentration of an analyte. For any particular detector, the relative response factor for the analyte compared to the internal standard must be determined first. Calibrating the linearity of the response factor for an analyte compared to the internal standard requires one to make a series of solutions with the same concentration of standard, and a varying concentration of analyte. A plot of the response of the analyte relative to the standard (peak area of analyte/peak area of standard) versus the concentration of the analyte relative to the standard ([analyte]/[standard]) should produce a linear response with a slope equal to the response factor, F. area of analyte signal concentration of analyte area of standard signal concentration of standard The table contains data for a chromatographic analysis of an analyte using an internal standard. Peak areas of each are measured by a mass spectrometer. The volume injected for Standard Analyte Standard Analyte concentration concentration peak (M) pea Sample (M) area area each sample is similar, but not exactly the same. 1.00 10.0 309 2991 Calculate the peak area ratio (analyte/standard) and the concentration ratio [analyte]/[standard] for each of the samples. Then, calculate F for each sample (peak area ratio/concentration ratio). 5.00 10.0 3677 6203 3 10.0 10.0 3025 2811
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Calculate the concentration of an aqueous solution of NaOH that has a pH of 11.50.
-
Calculate the concentration of an aqueous solution of Ca(OH)2 that has a pH of 10.05.
-
Calculate the concentration of HC 6 H 6 O 6 - in an aqueous solution of 0.0896 M ascorbic acid , H 2 C 6 H 6 O 6 (aq). [ HC 6 H 6 O 6 - ] = M.
-
a. Over a two year time horizon an investor experiences the following net cash flows: Year 0 1 2 Cash flow -$8,000 $5,280 $5,320 What is the internal rate of return for the investor? Demonstrate that...
-
Shay rolls a fair die until she gets a 6. If the random variable Y counts the number of times Shay rolls the die until she gets her first 6, determine (a) the probability distribution for F; (b)...
-
What will the code in Figure 4-54 assign to the lblStatus control when the strLevel variable contains the string 3? a. Bronze b. Gold c. Platinum d. Silver If strLevel = "1" OrElse strLevel "2" Then...
-
In July 2017, Latrice Merritt entered a residential lease with Doran 610 Apartments, LLC. Under the terms of the lease agreement, Merritt was prohibited from installing a private security system in...
-
Friendleys Miniature Golf and Driving Range Inc. was opened on March 1 by Dean Barley. These selected events and transactions occurred during March. Mar. 1 Stockholders invested $50,000 cash in the...
-
Lindal Corporation, organized in 2023, immediately filed an election for S corporation status under the rules of Subchapter S. What is the maximum amount of passive investment income that Lindal will...
-
The time-series graphs below show typical simulation output. For each graph identify the type of model (terminating or nonterminating) and the nature of the simulation output (transient,...
-
Abel Corp. manufactures a product that yields the by-product "Yum.". The only costs associated with Yum are selling costs of P.10 for each unit sold. Abel accounts for sales of Yum by deducting Yum's...
-
The probability that a live birth requires a cesarean delivery in a certain American state is 0.313. If 5 live births occur independently in this state, compute the probability that at least one of...
-
Describe a budget and how it is important in planning and control. Explain how small and large firms benefit from the budgeting process. Explain participative budgeting and some of its advantages and...
-
Required Information [The following Information applies to the questions displayed below.] Laker Company reported the following January purchases and sales data for its only product. For specific...
-
Suppose you want to move out of your apartment and buy a small house. Let s assume that your income is $ 3 9 , 6 0 0 a year, your student loans cost $ 2 5 0 per month, and your car payment is $ 3 2 5...
-
How do existentialist philosophies, such as existential authenticity and existential responsibility, inform the cultivation of authentic and morally resonant leadership that inspires profound...
-
Definitions of the standard cost card and the two types of standards Comparison of master budgets versus flexible budgets Definitions of volume variance and spending variance Responsible persons for...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
Consider the data presented in Exercise 14.19. (a) By using appropriate graphs, determine whether the reaction is first order or second order. (b) What is the rate constant for the reaction? (c) What...
-
What are the basic SI units for (a) The wavelength of light, (b) The frequency of light, (c) the speed of light?
-
How many nonbonding electron pairs are there in each of the following molecules: (a) (CH3)2S, (b) HCN, (c) H2C2, (d) CH3F?
-
Discretize the product of two functions, as compared to discretizing each function independently, and describe what that means in the language of kets.
-
Show how \(\delta^{\prime \prime}(x-y)\) (the second derivative with respect to \(x\) ) acts as a distribution on functions.
-
For a tensor product of kets, describe what the norm is in the abstract sense, and then in the function form (with integrals).

Study smarter with the SolutionInn App


