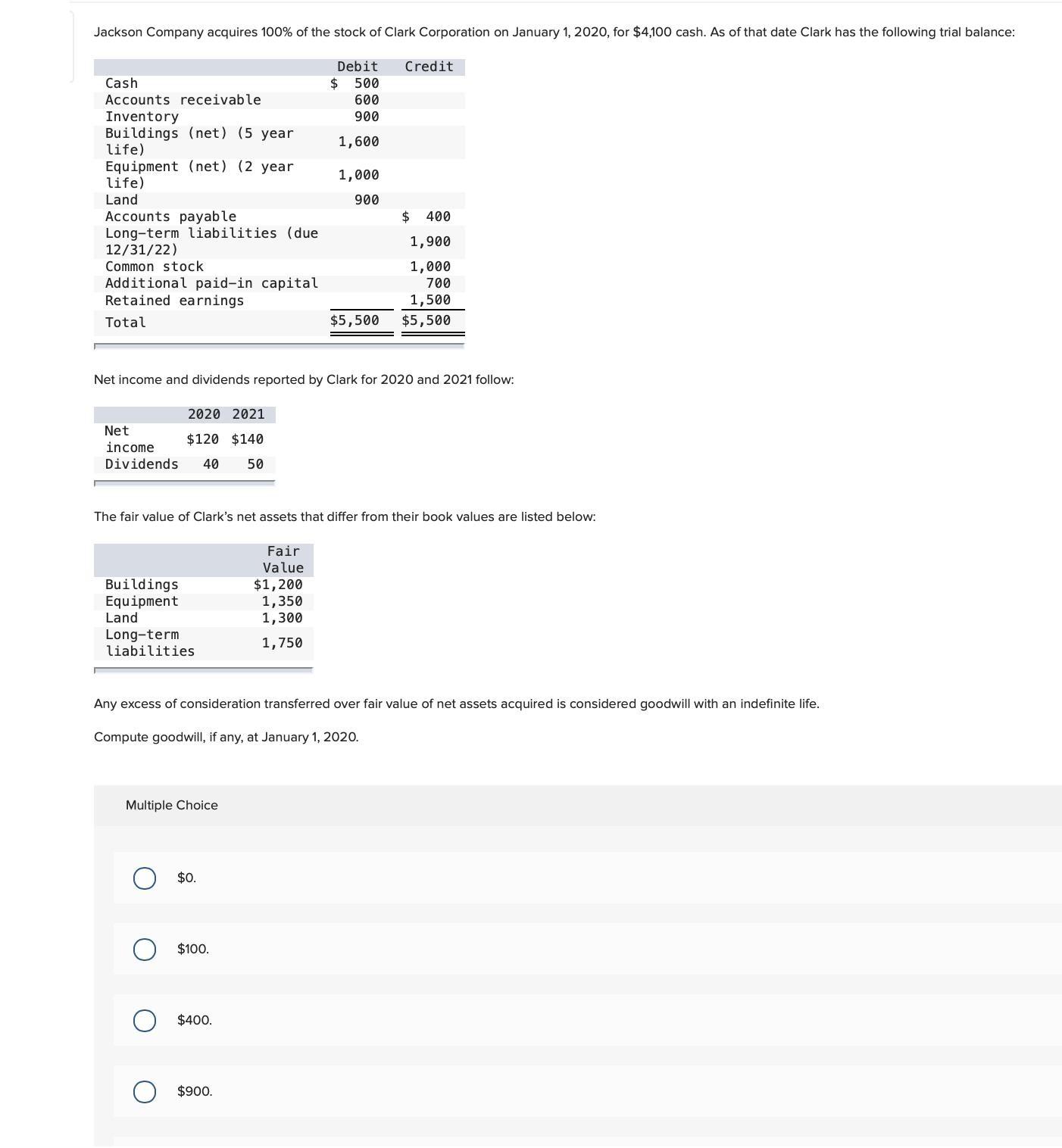
Jackson Company acquires 100% of the stock of Clark Corporation on January 1, 2020, for $4,100...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Jackson Company acquires 100% of the stock of Clark Corporation on January 1, 2020, for $4,100 cash. As of that date Clark has the following trial balance: Debit Credit Cash 24 500 Accounts receivable 600 Inventory Buildings (net) (5 year life) Equipment (net) (2 year life) 900 1,600 1,000 Land 900 $ 400 Accounts payable Long-term liabilities (due 12/31/22) 1,900 Common stock 1,000 700 Additional paid-in capital Retained earnings 1,500 Total $5,500 $5,500 Net income and dividends reported by Clark for 2020 and 2021 follow: 2020 2021 Net $120 $140 income Dividends 40 50 The fair value of Clark's net assets that differ from their book values are listed below: Fair Value Buildings Equipment Land $1,200 1,350 1,300 Long-term liabilities 1,750 Any excess of consideration transferred over fair value of net assets acquired is considered goodwill with an indefinite life. Compute goodwill, if any, at January 1, 2020. Multiple Choice $0. $100. $400. $900. Jackson Company acquires 100% of the stock of Clark Corporation on January 1, 2020, for $4,100 cash. As of that date Clark has the following trial balance: Debit Credit Cash 24 500 Accounts receivable 600 Inventory Buildings (net) (5 year life) Equipment (net) (2 year life) 900 1,600 1,000 Land 900 $ 400 Accounts payable Long-term liabilities (due 12/31/22) 1,900 Common stock 1,000 700 Additional paid-in capital Retained earnings 1,500 Total $5,500 $5,500 Net income and dividends reported by Clark for 2020 and 2021 follow: 2020 2021 Net $120 $140 income Dividends 40 50 The fair value of Clark's net assets that differ from their book values are listed below: Fair Value Buildings Equipment Land $1,200 1,350 1,300 Long-term liabilities 1,750 Any excess of consideration transferred over fair value of net assets acquired is considered goodwill with an indefinite life. Compute goodwill, if any, at January 1, 2020. Multiple Choice $0. $100. $400. $900.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Which of the following is taxable income? Rental income passed through by a partnership but not distributed to a cash basis partner Life insurance proceeds received by a closely-held corporation from...
-
On January 1 2020 Potter Company purchased 100 of the 1000 shares of Voldomort Company for $800. Potter has no significant influence over Voldomort On July 1, 2020, Voldomort declared and paid a $1...
-
The supplies account had a beginning balance of $1,523. Supplies purchased during the period totaled $4,928. At the end of the period before adjustment, $382 of supplies were hand. Prepare the...
-
A wire carries a current of 10 A in the +x-direction. (a) Find the force per unit length on the wire if it is in a magnetic field that has components of Bx = 0.020 T, By = 0.040 T, and Bz = 0 T. (b)...
-
A piece of insulated wire is shaped into a figure 8, as in Figure P31.18. The radius of the upper circle is 5.00 cm and that of the lower circle is 9.00 cm. The wire has a uniform resistance per unit...
-
It is generally known that boys grow at an unusually fast rate between the ages of about 12 and 14. Following are heights, in inches, of 40 boys measured at age 12 and again at age 14. Can you...
-
The following merchandise transactions occurred in December. Both companies use a perpetual inventory system. Dec. 3 Pippen Company sold merchandise to Thomas Co. for $32,000, terms 2/10, n/30, FOB...
-
A prospective borrower has an estimated monthly housing expense of$ 1 , 6 0 0 and his monthly obligations total $ 2 , 1 0 0 . If the borrower\'s monthly grossincome is $ 5 , 8 4 5 , what is the total...
-
Use the following forward and spot prices for Canadian dollars (C$) to answer the question below. The prices are in U.S. dollars ($/C$). Forward rate for 4/15/20 Spot rate delivery of Canadian...
-
Your client, Jill, has a rapidly growing marketing business. She's filing single and projecting a profit of $500,000 for her business, a Single Member LLC which is filing on Schedule C. Her taxable...
-
How would I make this program run without the use of an array class /********************************************************************************************** *Description: CatchThief is a...
-
Cleanteq Metal Recovery, which is specifically established for a joint venture agreement to extract gold, will end its corporate life in 3 years. Net Cash Flow expected during the years it still...
-
Siegfried, 67, has a significant cash surrender value (CSV) built up in the life insurance policy he purchased on December 31, 1982. He requires cash for living expenses and has turned to you for...
-
Jesse Derr formed a corporation to conduct his construction business. Although the actual corporate name was JCDER Inc., Der regularly referred to the corporation as J.D Construction company. In...
-
Tax Consequences. Katie paid $9,200 for a Ginnie Mae bond with a par value of $10,000 and a coupon rate of 6.7%. Two years later, after having received the annual interest payments on the bond, Katie...
-
Golf Unlimited Inc. (GUI), an entity that reports under ASPE, acquired a building at a cost of $1,000,000 on January 1, Year 1. Amortization for the building was set on a straight-line basis over 25...
-
Difference between truncate & delete
-
You are the credit manager for Meyer Company. One of your sales staff has made a $60,000 credit sale to Rudy Technology, a manufacturer of small computers. Your responsibility is to decide whether to...
-
Refer to the information for Morgan Inc. above. If Morgan uses a periodic inventory system, what is the cost of ending inventory under average cost at April 30? (Use four decimal places for per-unit...
-
From time to time, business news will report that the management of a company has misstated its profits by knowingly establishing an incorrect amount for its ending inventory . Required: 1. Explain...
-
A hydrocarbon \(\mathrm{H}_{2}\left(\mathrm{CH}_{2} ight)_{n}\) is dissolved in a solvent \(S\) which freezes at \(9.0^{\circ} \mathrm{C}\). A solution which contains \(0.90 \mathrm{~g}\) of...
-
Estimate the depression in the freezing point if \(58.5 \mathrm{~g}\) of \(\mathrm{NaCl}\) is added to \(1 \mathrm{~L}\) of water at atmospheric pressure.
-
Calculate the mass of methyl alcohol which, when dissolved in \(100 \mathrm{~g}\) of water, would just prevent the formation of ice at \(-10^{\circ} \mathrm{C}\), given that \(K_{\mathrm{f}}\) is...

Study smarter with the SolutionInn App


