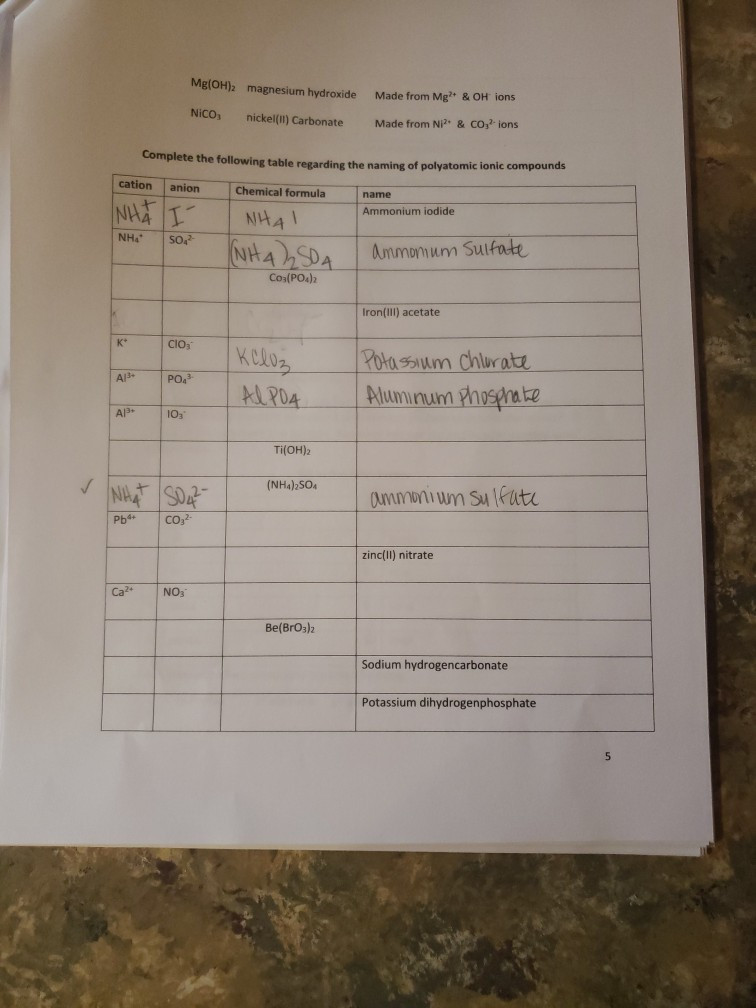
K* Complete the following table regarding the naming of polyatomic ionic compounds cation anion Chemical formula...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
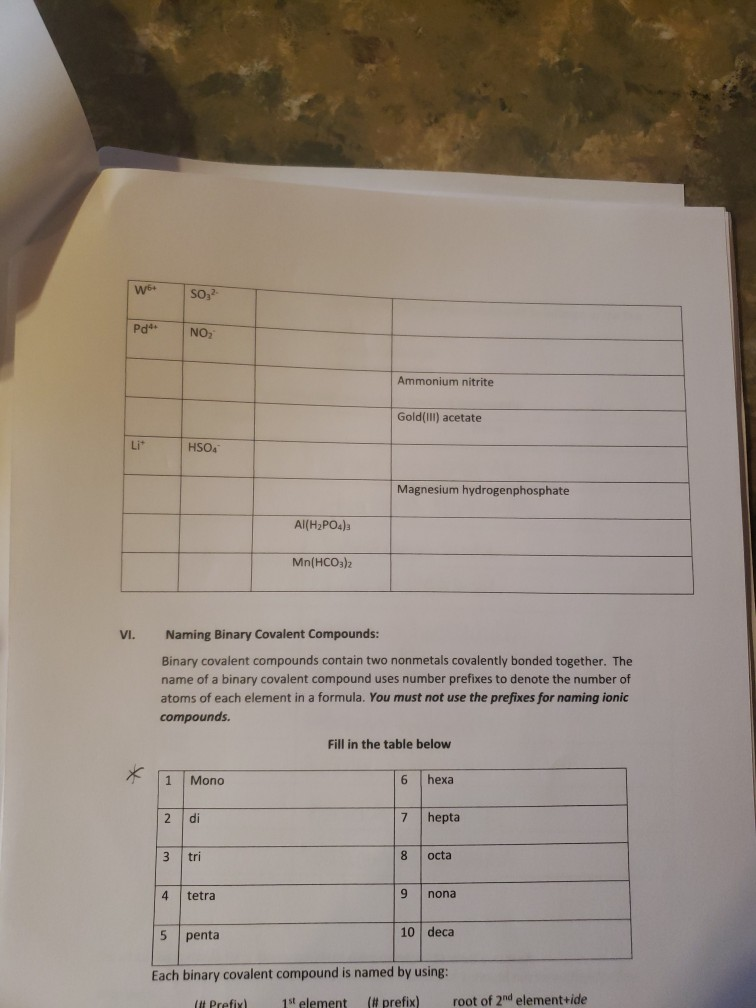
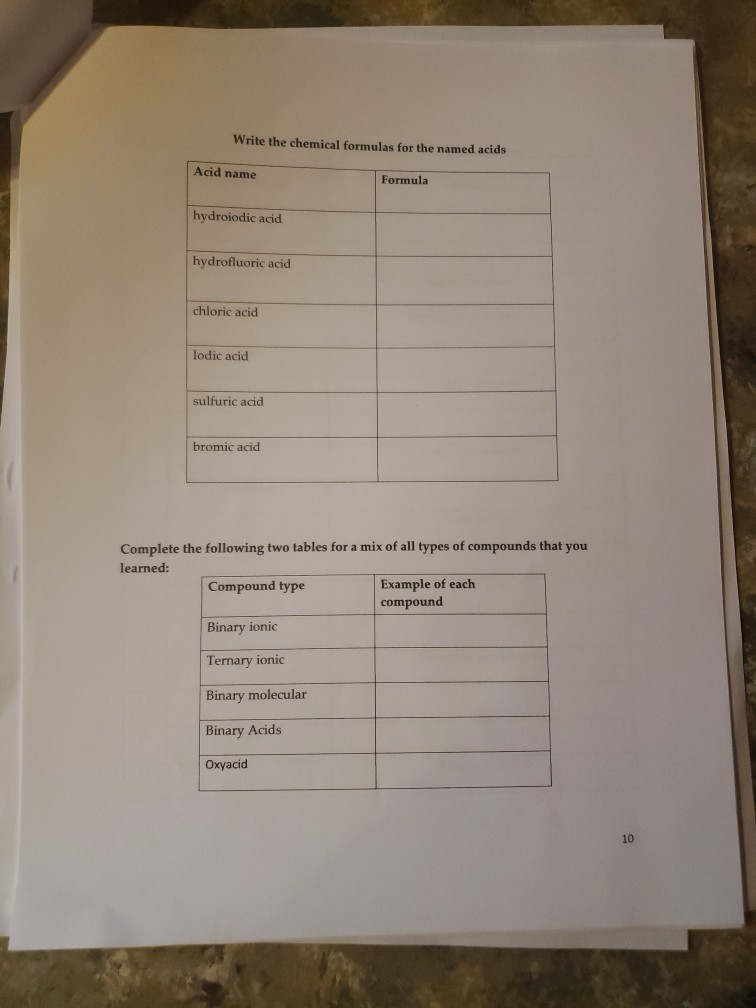
✓ K* Complete the following table regarding the naming of polyatomic ionic compounds cation anion Chemical formula NHÀ Í NH41 NH₂ NHASDA CO₂(PO4)2 Al3+ Al³+ Ca²+ Mg(OH)2 magnesium hydroxide NICO₁ nickel(II) Carbonate SO4²- CIOs PO4³ 10₂₁ NHÀ SO Pb4+ CO32- NO₁™ кслоз AlPO4 Ti(OH)2 (NH4)2SO4 Made from Mg2+ & OH ions Made from Ni²+ & CO3²- ions Be(BrO3)2 name Ammonium iodide Ammomum Sulfate Iron(III) acetate Potassium Chlorate Aluminum Phosphate ammonium sulfate zinc(II) nitrate Sodium hydrogencarbonate Potassium dihydrogenphosphate W5+ Pd4 Li VI. SO₂²- 1 NO₂ HSOA Mono 2 di 3 tri Naming Binary Covalent Compounds: Binary covalent compounds contain two nonmetals covalently bonded together. The name of a binary covalent compound uses number prefixes to denote the number of atoms of each element in a formula. You must not use the prefixes for naming ionic compounds. 4 tetra Al(H₂PO4)3 5 penta Mn(HCO3)2 Ammonium nitrite Gold(III) acetate Magnesium hydrogenphosphate Fill in the table below 6 7 8 9 hexa hepta octa nona 10 deca Each binary covalent compound is named by using: ( Prefix) 1st element (# prefix) root of 2nd element+ide Write a formula for the following binary covalent compounds: carbon disulfide sulfur trioxide dinitrogen pentoxide trisilicon tetrahydride sulfur hexafluoride Trisulfur heptanitride VII. Naming molecular acids: Acids are uncharged molecules. They contain the familiar anions with added H* ions. Naming Binary Acids: Binary acids (two atoms types only, hydrogen + a nonmetal) are named by adding hydro at the beginning of the respective anion name and the end of the anion name is changed to ic acid from ide. Ex. HCl, a binary acid is named: hydrochloric acid (respective anion is chloride [Cl]). Naming ternary oxyacids: Oxyacids are ternary (3 elements). They contain oxygen, hydrogen and another element. If the anion is an oxyanion then change the ate ending of the polyatomic anion that they contain to ic acid. A prefix is not needed. Most molecular ions that you have learnt about have an oxygen in them and they are therefore oxyanions. Ex. HNO3 is named: Nitric acid (respective anion is nitrate [NO3]). Continue to next page Write the chemical formulas for the named acids Acid name hydroiodic acid hydrofluoric acid chloric acid Iodic acid sulfuric acid bromic acid Complete the following two tables for a mix of all types of compounds that you learned: Compound type Formula Binary ionic Ternary ionic Binary molecular Binary Acids Oxyacid Example of each compound 10 Name S/N 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Chemical Formula SinCh H₂O₂ COS SnO₂ Cds(PO4)2 Zn3P₂ Pb(OH)2 H₂SO4 SnP₂ Section Time_ Name Beryllium iodide Triphosphorus hexanitride Gold(III) carbonate Copper (1) hydroxide Silver nitride Nitric acid Tetrasulfur pentachloride Aluminum nitride 11 18 19 20 21 22 23 24 25 26 27 28 29 30 31 33 34 35 36 37 38 Fe (SO4)3 NISO, Cu(NO3)2 НЕ Cd(CH3COO)2 Pb(BrO3)2 KNO InP MnSe₂ V₂05 Zinc(11) hydrogencarbonate Copper (1) sulfide Chromium(VI) dihydrogenphosphate Phosphoric acid Iron(III) carbonate Acetic acid Cobalt(II) chlorite Barium Nitrite Strontium phosphate Titanium(II) bromide 12 ✓ K* Complete the following table regarding the naming of polyatomic ionic compounds cation anion Chemical formula NHÀ Í NH41 NH₂ NHASDA CO₂(PO4)2 Al3+ Al³+ Ca²+ Mg(OH)2 magnesium hydroxide NICO₁ nickel(II) Carbonate SO4²- CIOs PO4³ 10₂₁ NHÀ SO Pb4+ CO32- NO₁™ кслоз AlPO4 Ti(OH)2 (NH4)2SO4 Made from Mg2+ & OH ions Made from Ni²+ & CO3²- ions Be(BrO3)2 name Ammonium iodide Ammomum Sulfate Iron(III) acetate Potassium Chlorate Aluminum Phosphate ammonium sulfate zinc(II) nitrate Sodium hydrogencarbonate Potassium dihydrogenphosphate W5+ Pd4 Li VI. SO₂²- 1 NO₂ HSOA Mono 2 di 3 tri Naming Binary Covalent Compounds: Binary covalent compounds contain two nonmetals covalently bonded together. The name of a binary covalent compound uses number prefixes to denote the number of atoms of each element in a formula. You must not use the prefixes for naming ionic compounds. 4 tetra Al(H₂PO4)3 5 penta Mn(HCO3)2 Ammonium nitrite Gold(III) acetate Magnesium hydrogenphosphate Fill in the table below 6 7 8 9 hexa hepta octa nona 10 deca Each binary covalent compound is named by using: ( Prefix) 1st element (# prefix) root of 2nd element+ide Write a formula for the following binary covalent compounds: carbon disulfide sulfur trioxide dinitrogen pentoxide trisilicon tetrahydride sulfur hexafluoride Trisulfur heptanitride VII. Naming molecular acids: Acids are uncharged molecules. They contain the familiar anions with added H* ions. Naming Binary Acids: Binary acids (two atoms types only, hydrogen + a nonmetal) are named by adding hydro at the beginning of the respective anion name and the end of the anion name is changed to ic acid from ide. Ex. HCl, a binary acid is named: hydrochloric acid (respective anion is chloride [Cl]). Naming ternary oxyacids: Oxyacids are ternary (3 elements). They contain oxygen, hydrogen and another element. If the anion is an oxyanion then change the ate ending of the polyatomic anion that they contain to ic acid. A prefix is not needed. Most molecular ions that you have learnt about have an oxygen in them and they are therefore oxyanions. Ex. HNO3 is named: Nitric acid (respective anion is nitrate [NO3]). Continue to next page Write the chemical formulas for the named acids Acid name hydroiodic acid hydrofluoric acid chloric acid Iodic acid sulfuric acid bromic acid Complete the following two tables for a mix of all types of compounds that you learned: Compound type Formula Binary ionic Ternary ionic Binary molecular Binary Acids Oxyacid Example of each compound 10 Name S/N 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Chemical Formula SinCh H₂O₂ COS SnO₂ Cds(PO4)2 Zn3P₂ Pb(OH)2 H₂SO4 SnP₂ Section Time_ Name Beryllium iodide Triphosphorus hexanitride Gold(III) carbonate Copper (1) hydroxide Silver nitride Nitric acid Tetrasulfur pentachloride Aluminum nitride 11 18 19 20 21 22 23 24 25 26 27 28 29 30 31 33 34 35 36 37 38 Fe (SO4)3 NISO, Cu(NO3)2 НЕ Cd(CH3COO)2 Pb(BrO3)2 KNO InP MnSe₂ V₂05 Zinc(11) hydrogencarbonate Copper (1) sulfide Chromium(VI) dihydrogenphosphate Phosphoric acid Iron(III) carbonate Acetic acid Cobalt(II) chlorite Barium Nitrite Strontium phosphate Titanium(II) bromide 12
Expert Answer:
Answer rating: 100% (QA)
Cation Anion Chemical Formulae Name NH4 I NH4I Ammoniu... View the full answer

Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Posted Date:
Students also viewed these chemistry questions
-
If the nominal rate of return is 11.09% and the inflation rate is 4.62%, by what percent is actual purchasing power increasing? Enter your answer as a percentage with four decimal places but without...
-
Complete the following table showing which ledger account is to be debited and which is to be credited: Debit Credit al Sratorery purchased on credit from Gomley bl Rates paid by direct debit...
-
Zannel plc is considering a new project to produce a revolutionary surveillance device. The initial capital costs of 300,000 will be paid immediately. The project is expected to last five years. The...
-
Compute the determinant below. 14 8 8 8 51 1000 8 7 3 0 0 0 8 8 8 8 8 2
-
Kendra, Cogley, and Mei share income and loss in a 3:2:1 ratio. The partners have decided to liquidate their partnership. On the day of liquidation their balance sheet appears as follows. Required...
-
Mahendra started a business on 1 November 207. He provided the following information for his first month of trading: Nov 1 Introduced $160 000 capital into the business, of which $158 500 was...
-
Virtuoso Transportation issued \(\$ 600,000\) of \(8 \%\) bonds payable at \(9^{-}\) on October 1, 2010. These bonds are callable at 100 and mature on October 1, 2018. Virtuoso pays interest each...
-
Jay-Zee Company makes an in-car navigation system. Next year, Jay-Zee plans to sell 16,000 units at a price of $320 each. Product costs include: Direct materials ....... $68 Direct labor .............
-
You are trying to choose among three investments described below: Investment A: Up-front investment of $45,000 and returns $120,000 in six years. Investment B: Up-front investment of $60,000 and...
-
Kennedy Prisby, age 33, is single and has no dependents. Her social security number is 111-22-3333. Her address is 101 North Fork Ave., Cedar City, UT 84720. She has no intention of donating to the...
-
Write the function strip(s) that takes in a string and returns the same string with any leading and trailing spaces removed. For this problem, we are only considering " " as a space. we cannot use...
-
You are expected to find 1 current news article from a credible media NEWS source (current Canadian newspaper, magazine) and analyze the contents. Summary - should be in a paragraph, outlining the...
-
The following five tasks are to be scheduled on a single processor preemptively. Construct the schedule when the priority ceiling protocol is applied for resource access control. Explain the schedule...
-
Date range: 1 June 2018 - 8 June 2018 Bank reconciliation is the process of matching bank statement lines imported into Xero from your bank account, with account transactions you've created in Xero...
-
Think aboutan experience you hadwith another culture; or you may describe what happened to someone you know. A PowerPoint presentation detailing the following: What werethe impressions, both positive...
-
From 2021-2023, many industries across the world faced a shortage of computer chips. In particular, the auto industry faced a shortage of key chips needed to manufacture cars. Below is hypothetical...
-
Find the IQR interquartile range of the following data set 12,16,14,13,15.8,18,22,5.
-
If someone's Z-score for a variable was 0.67. Their score is a significant extreme score. Their score is not significant. O Their score is slightly above average. O Their score is an outlier.
-
Ammonia, NH 3 , reacts with chlorine gas, Cl 2 , to form ammonium chloride, NH 4 Cl, and nitrogen trichloride, NCl 3 . Write a balanced equation for this reaction.
-
What gave Schrdinger justification to think of an electron in an atom as a nebulous cloud?
-
Mendeleev arranged the elements by atomic mass and then separated them into various groups. What was his criterion for which elements were grouped together?
-
Ten students are chosen from a statistics class of 25 students. Let X be the number who got an A in the class. In Exercises 1116, determine whether the random variable X has a binomial distribution....
-
In a college with 5000 students, 100 are randomly chosen to complete a survey in which they rate the quality of the cafeteria food. Let X be the number of freshmen who are chosen. Does X have a...
-
= 0.1, t = 10, P(2) In Exercises 918, determine the indicated probability for a Poisson random variable with the given values of and t.

Study smarter with the SolutionInn App


