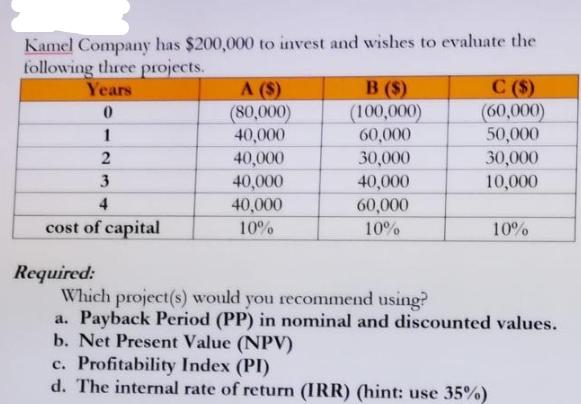
Kamel Company has $200,000 to invest and wishes to evaluate the following three projects. Years 0...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Kamel Company has $200,000 to invest and wishes to evaluate the following three projects. Years 0 1 2 3 4 cost of capital Required: A ($) (80,000) 40,000 40,000 40,000 40,000 10% B ($) (100,000) 60,000 30,000 40,000 60,000 10% C($) (60,000) 50,000 30,000 10,000 10% Which project(s) would you recommend using? a. Payback Period (PP) in nominal and discounted values. b. Net Present Value (NPV) c. Profitability Index (PI) d. The internal rate of return (IRR) (hint: use 35%) Kamel Company has $200,000 to invest and wishes to evaluate the following three projects. Years 0 1 2 3 4 cost of capital Required: A ($) (80,000) 40,000 40,000 40,000 40,000 10% B ($) (100,000) 60,000 30,000 40,000 60,000 10% C($) (60,000) 50,000 30,000 10,000 10% Which project(s) would you recommend using? a. Payback Period (PP) in nominal and discounted values. b. Net Present Value (NPV) c. Profitability Index (PI) d. The internal rate of return (IRR) (hint: use 35%)
Expert Answer:
Answer rating: 100% (QA)
To evaluate the projects and determine which ones to recommend we will calculate the payback period net present value NPV profitability index PI and i... View the full answer

Related Book For 

Applied Regression Analysis and Other Multivariable Methods
ISBN: 978-1285051086
5th edition
Authors: David G. Kleinbaum, Lawrence L. Kupper, Azhar Nizam, Eli S. Rosenberg
Posted Date:
Students also viewed these accounting questions
-
Find 2 different advertisements for jobs in your field or the field you desire to get into. List the job and the company and why you want to work there. What's available? How much is the salary? and...
-
Ques 1: What is the major complaint by firms concerning the Sarbanes-Oxley act of 2012? A. the legislative maximum allowable compensation for a CEO. B. the legal requirement to disclose project...
-
The net present value and internal rate of return desirability measures for two mutually exclusive investments being considered by Stockton Corporation to follow. Year NPV IRR R 161 14.60% S 138...
-
O 00:29:33 4. Let an be a convergent series and b, be a soquence such that 0 <2+ a, < b Which of the following statements are true? 10 I) The convergence or divergence of cannot be concluded. Im (2+...
-
A certain parallel-plate capacitor is filled with a dielectric for which K = 5.5. The area of each plate is 0.034 m2, and the plates are separated by 2.0 mm. The capacitor will fail (short out and...
-
Using Program15.m (central difference method), solve Problem 11.18 with \(c=1\). Data From Problem 11.18:- Using the central difference method, find the response of the two-degree-of-freedom system...
-
When testing several hypotheses, it is reasonable to reject every hypothesis for which P < 0.05. In Exercises 5 and 6, determine whether the statement is true or false. If the statement is false,...
-
Yarnell Electronics sells computer systems to small businesses. Yarnell engaged in the following activities involving notes receivable: a. On November 1, 2011, Yarnell sold a $5,000 system to Ross...
-
TRD Company is expected to generate free cash flow to equity holders of $2.0 million, 2.5 Million, and 3.0 million in 2021,2022,2023. Beyond 2023, TRD expects to grow free cash flows to equity...
-
Determine the hydrostatic force F on the triangular gate, which is hinged at the bottom edge and held by the reaction RT at the upper corner. Express F in terms of , h, and W. Also determine the...
-
A local entrepreneur, Frodo Baggins, buys old computers, fixes them, and resells them to the public. Create excel formulas that will calculate sale price of each car that includes the Fredo's cost,...
-
Write a test client for Rectangle that takes three command-line arguments \(\mathrm{n}\), min, and max; generates \(\mathrm{n}\) random rectangles whose width and height are uniformly distributed...
-
Are you optimistic or pessimistic about the future? Why?
-
Would the export of hazardous waste to developing countries be efficient? Sometimes? Always? Never? Would it be moral? Sometimes? Always? Never? Make clear the specific reasons for your judgments.
-
Future generations can cast neither votes in current elections nor dollars in current market decisions. Therefore, it should not come as a surprise to anyone that the interests in future generations...
-
Fertility rates vary widely among various ethnic groups in the United States. Black and Spanish-speaking Americans have above-average rates, for example, while Jews have below-average fertility...
-
Q1 a) Find the Present Values of the following Ordinary Annuities: $800 per year for 10 years at 10% $400 per year for 5 years at 5% $500 per year for 6 years at 8% i. ii. iii. b) Find the Present...
-
Use the method of Example 4.29 to compute the indicated power of the matrix. 1 0 1
-
A company wants to compare three different point-of-sale promotions for its snack foods. The three promotions are Promotion 1: Buy two items, get a third free. Promotion 2: Mail in a rebate for $1.00...
-
Consider hypothetical data based on a study concerning the effects of rapid cultural change on blood pressure levels for native citizens of an island in Micronesia. Blood pressures were taken on a...
-
Consider the numerical examples given in Section 8.8 of Chapter 8, involving assessment of the relationship of the independent variables HGT, AGE, and (AGE)2 to the dependent variable WGT. Suppose...
-
When there is a concentration gradient in the system, show that the potential gradient is composed of two terms, (i) an Ohm's-law contribution and (ii) a diffusional contribution. State the equation...
-
Copper is deposited at a cathode from solution with a bulk concentration of \(0.5 \mathrm{M}\) at the rate of \(3.0 \mathrm{~g} / \mathrm{m}^{2} \cdot \mathrm{s}\). Find the surface concentration of...
-
Find the mobility of \(\mathrm{H}^{+}, \mathrm{OH}^{-}\), and other ions from the diffusivity data given in Table 22.1. Table 22.1. Diffusion coefficients of ions in water at 25 C. H+ Na+ K+ Ca+ OH-...

Study smarter with the SolutionInn App


