Question: Draw the structure for the product of the following reaction. If more than one product can reasonably be conceived from the reaction, draw the
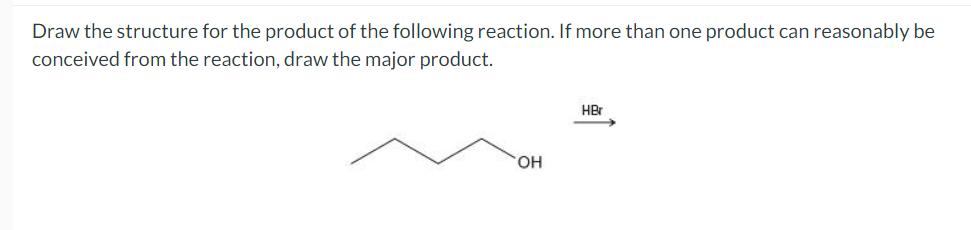
Draw the structure for the product of the following reaction. If more than one product can reasonably be conceived from the reaction, draw the major product. OH HBr
Step by Step Solution
3.33 Rating (147 Votes )
There are 3 Steps involved in it
The product of the reaction HEW OH is methanol CH3OH HEW or hydroxy... View full answer

Get step-by-step solutions from verified subject matter experts


