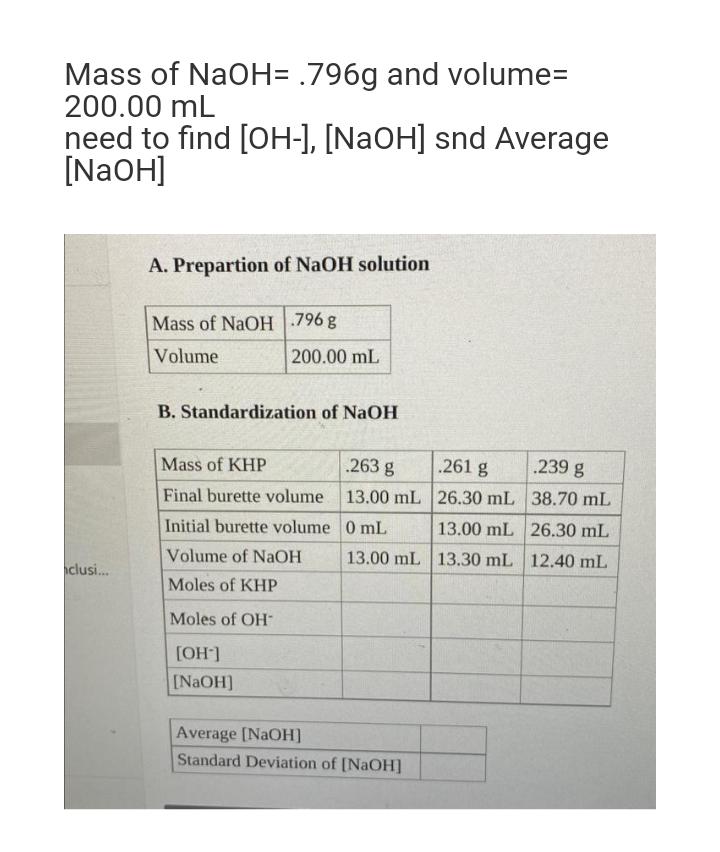
Mass of NaOH= .796g and volume= 200.00 mL need to find [OH-], [NaOH] snd Average [NaOH]...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Mass of NaOH= .796g and volume= 200.00 mL need to find [OH-], [NaOH] snd Average [NaOH] clusi... A. Prepartion of NaOH solution Mass of NaOH 796 g Volume 200.00 mL B. Standardization of NaOH Mass of KHP Final burette volume Initial burette volume Volume of NaOH Moles of KHP Moles of OH [OH-] [NaOH] .263 g 13.00 mL 0 mL 13.00 mL Average [NaOH] Standard Deviation of [NaOH] .261 g 26.30 mL 13.00 mL 13.30 mL .239 g 38.70 mL 26.30 mL 12.40 mL Mass of NaOH= .796g and volume= 200.00 mL need to find [OH-], [NaOH] snd Average [NaOH] clusi... A. Prepartion of NaOH solution Mass of NaOH 796 g Volume 200.00 mL B. Standardization of NaOH Mass of KHP Final burette volume Initial burette volume Volume of NaOH Moles of KHP Moles of OH [OH-] [NaOH] .263 g 13.00 mL 0 mL 13.00 mL Average [NaOH] Standard Deviation of [NaOH] .261 g 26.30 mL 13.00 mL 13.30 mL .239 g 38.70 mL 26.30 mL 12.40 mL
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A chunk of ice of mass ml = 100 g at a temperature tx = 0 C was placed in a calorimeter in which water of mass m1 = 100 g was at a temperature t1. Assuming the heat capacity of the calorimeter to be...
-
A gas has an initial volume of 638 mL and an initial pressure of 779 torr. What is its final volume in liters if its pressure is changed to 0.335 atm? Assume temperature and amount are held constant.
-
A gas has an initial volume of 685 mL and an initial temperature of 29C. What is its new temperature if volume is changed to 1.006 L? Assume pressure and amount are held constant.
-
The membrane filter technique is used to test a polluted water sample for coliform group. Three different dilutions of the water sample were prepared and each was filtered through 5 filter membranes....
-
A coach knows it is vital that the volleyballs be fully inflated before a match. (a) Calculate the impulse on a spiked 0.123-slug volleyball when the incoming velocity of the ball is 11.5 ft/s and...
-
In Problems 7 18, write the augmented matrix of the given system of equations. x+y - z = 2 3x-2y = 2 5x+3y-z= 1 =
-
Use the information from the balance sheet and income statement below to calculate the following ratios: Current ratio Acid-test ratio Times interest earned Inventory turnover Total asset turnover...
-
The following transactions of Plymouth Pharmacies occurred during 2011 and 2012: Requirement 1. Journalize the transactions in Plymouths general journal. Explanations are notrequired. 2011 Purchased...
-
10. A skier plans to ski a smooth fixed hemisphere of radius R. He starts form rest on a curved smooth surface of height TR/4 The angle at which he leaves the hemisphere is : (a) cos Yshipma (b) cos...
-
The following relate to auditors independence: a. Why is independence so essential for auditors? b. Compare the importance of independence of CPAs with that of other professionals, such as attorneys....
-
17. Suppose that during the TCP connection closing procedure, a machine that is in the TIME WAIT state crashes, reboots within MSL seconds, and immediately attempts to reestablish the connection...
-
If an item purchased at a marked-up price from a group company in the previous year is resold in the current year, group gross profit will increase and group beginning retained earnings will reduce....
-
A profit from an intercompany sale would result in either a higher cost of sales in the income statement or a higher inventory balance in the statement of financial position of the buying company....
-
Dividend income in the profit or loss of a parent company should be eliminated against dividend payable in the statement of financial position of a subsidiary company. True/False
-
Payables and receivables in the group statement of financial position include balances with associate companies. True/False
-
An acquirer is called a parent when it acquires the net assets rather than the equity of another entity, called the subsidiary. Is this true or false?
-
A drug store (pharmacy) chain wants to raise $500,000 using two-month commercial paper. The net proceeds to the chain will be $497,500. What is the effective annual rate of this financing? Given what...
-
Software Solution is family-owned business that has been in operation for more than 15 year. The board of directors is comprised of mainly family members, plus a few professionals such as an...
-
Write a balanced equation for the thermal decomposition of aluminum nitrate to form aluminum oxide, nitrogen dioxide, and oxygen gas.
-
Four atoms are arbitrarily labeled D, E, F, and G. Their electro negativities are as follows: D = 3.8, E = 3.3, F = 2.8, and G = 1.3. If the atoms of these elements form the molecules DE, DG, EG, and...
-
Platinum forms two different compounds with chlorine. One contains 26.7 percent Cl by mass, and the other contains 42.1 percent Cl by mass. Determine the empirical formulas of the two compounds.
-
Apple stock is selling for \($120\) per share. Call options with a \($117\) exercise price are priced at \($12.\) What is the intrinsic value of the option, and what is the time value?
-
Name five variables that can affect the price of options, and briefly explain how each affects prices. How important are intrinsic value and time value to in-the-money options? To out-of-the-money...
-
Twitter is trading at \($34.50.\) Call options with a strike price of \($35\) are priced at \($2.30\) . What is the intrinsic value of the option, and what is the time value?

Study smarter with the SolutionInn App



