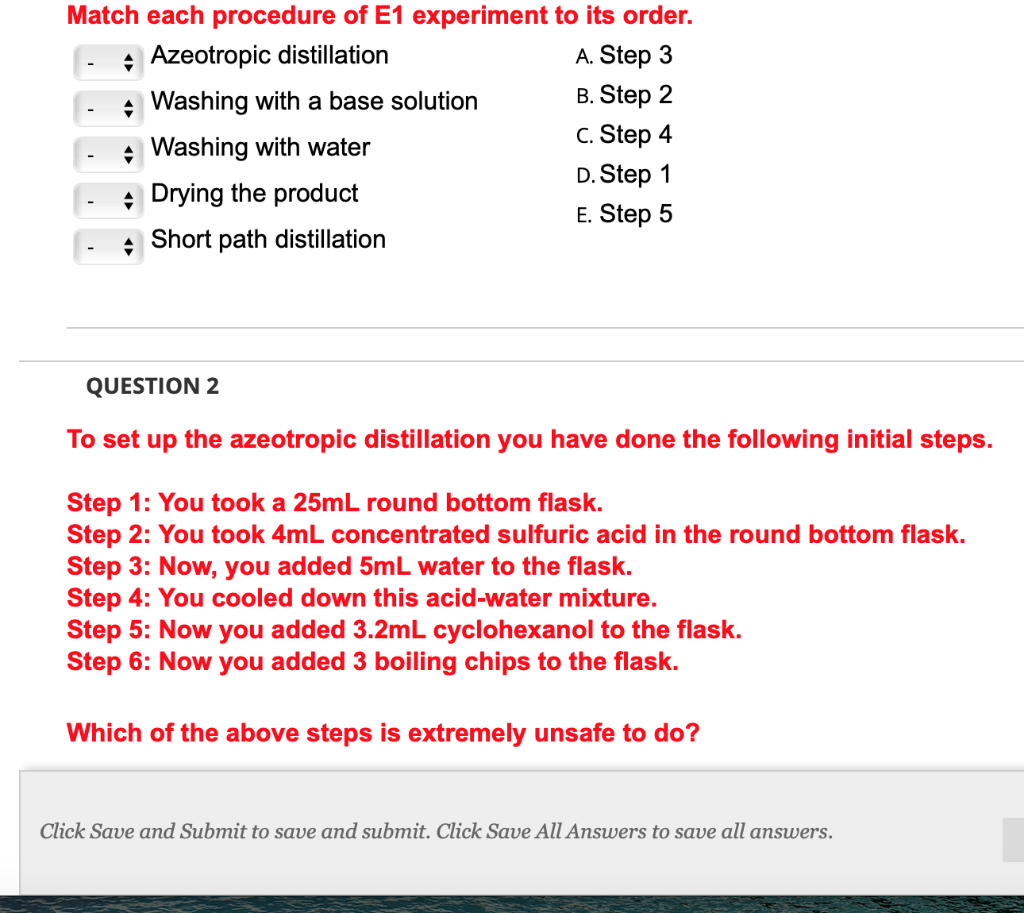
Match each procedure of E1 experiment to its order. Azeotropic distillation A. Step 3 Washing with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Match each procedure of E1 experiment to its order. Azeotropic distillation A. Step 3 Washing with a base solution B. Step 2 Washing with water c. Step 4 D. Step 1 Drying the product E. Step 5 Short path distillation QUESTION 2 To set up the azeotropic distillation you have done the following initial steps. Step 1: You took a 25mL round bottom flask. Step 2: You took 4mL concentrated sulfuric acid in the round bottom flask. Step 3: Now, you added 5mL water to the flask. Step 4: You cooled down this acid-water mixture. Step 5: Now you added 3.2mL cyclohexanol to the flask. Step 6: Now you added 3 boiling chips to the flask. Which of the above steps is extremely unsafe to do? Click Save and Submit to save and submit. Click Save All Answers to save all answers. Match each procedure of E1 experiment to its order. Azeotropic distillation A. Step 3 Washing with a base solution B. Step 2 Washing with water c. Step 4 D. Step 1 Drying the product E. Step 5 Short path distillation QUESTION 2 To set up the azeotropic distillation you have done the following initial steps. Step 1: You took a 25mL round bottom flask. Step 2: You took 4mL concentrated sulfuric acid in the round bottom flask. Step 3: Now, you added 5mL water to the flask. Step 4: You cooled down this acid-water mixture. Step 5: Now you added 3.2mL cyclohexanol to the flask. Step 6: Now you added 3 boiling chips to the flask. Which of the above steps is extremely unsafe to do? Click Save and Submit to save and submit. Click Save All Answers to save all answers.
Expert Answer:
Answer rating: 100% (QA)
Azeotropic distillation C Step 4 Washing with a base solution B Step 2 Was... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Concentrated sulfuric acid reacts with sodium iodide to produce molecular iodine, hydrogen sulfide, and sodium hydrogen sulfate. Write a balanced equation for the reaction.
-
A weak acid is added to a concentrated solution of hydrochloric acid. Does the solution become more or less acidic?
-
The concentration of commercially available concentrated sulfuric acid is 98.0 percent by mass, or 18 M. Calculate the density and the molality of the solution.
-
A fast-food restaurant operates both a drive through facility and a walk-in facility. On a randomly selected day, let X and Y, respectively, be the proportions of the time that the drive-through and...
-
Acme Bricks, a masonry products company, wants to have $600,000 on hand before it invests in new conveyors, trucks, and other equipment. If the company sets aside $80,000 per year in an account that...
-
What is the difference between the database design scope and its boundaries? Why is the scope and boundary statement so important to the database designer?
-
When a party may be compelled to defend an action in a particular court?
-
Satu Company, a merchandiser, recently completed its 2013 operations. For the year, (1) all sales are credit sales, (2) all credits to Accounts Receivable reflect cash receipts from customers, (3)...
-
explain how does intermarket swap work? why when the yield spread gets smaller, the corporate bonds will outperform the treasury bonds? Active Bond Investment Basis: you can predict interest rate...
-
Telstar uses job order costing. The T-accounts below summarize its production activity for the year. 1. Compute the amount for each of the following. a. Direct materials used b. Indirect materials...
-
7. Consider the figure below. HO -C pka-COOH-2.19 pka-NH2 = 9.67 pka-sidechain 4.25 CHCH2C OH a. What amino acid is this? (1) b. Is it in the R or S configuration? (2) c. Draw the three forms of the...
-
What is community psychology (CP)?
-
From the reading Welcome to Psychology 2e , an OpenStax in what ways do you have a better understanding of psychology? give examples and cite from the text
-
Roberta Bondar is travelling in her space craft around a distant planet. The planet has a radius R' and she is at an altitude of 2R' above its surface, coasting at some constant speed in a circular...
-
Morten Hansen, in his book Great at Work , challenges the long-held practice and belief by many organizations that in order to succeed and grow the organization, employees need to work longer hours....
-
The Sanberg Company had 331 units on hand at the beginning of the year, with a unit cost of $5.80. The number of units purchased and the unit cost and the number of units sold during the year are...
-
For the system below draw the signal flow graph and hence derive the transfer function. Find the steady state error to a step input R(s) 3 s+2 y(s)
-
Suppose you won a financial literacy competition and are given FJS10000 to invest, with the condition that investment can be done either in, i) Invest in Unit trust of Fiji or Invest in Fijian...
-
In 2012, Margaret and John Murphy are married taxpayers who file a joint tax return with AGl of $25,000. During the year they incurred the following expenses: Hospitalization insurance...
-
Diane is a single taxpayer who qualifies for the earned income credit. Diane has two qualifying children who are 3 and 5 years old. During 2012, Diane's wages are $17,100 and she receives dividend...
-
Olive Corporation was formed and began operations on January 1, 2012. The corporation's income statement for the year and the balance sheet at year-end are presented below. The corporation made...
-
Find an example of a large project that took more than a year to complete. You can ask people at your college, university, or work about a recent project, such as a major fundraising campaign,...
-
What is the main purpose of a project quality management plan? What are two metrics besides those provided in this chapter that Kristin and her team could use on the Just-In-Time Training project?
-
Your organization initiated a project to raise money for an important charity. Assume that there are 1,000 people in your organization. Also, assume that you have six months to raise as much money as...

Study smarter with the SolutionInn App


