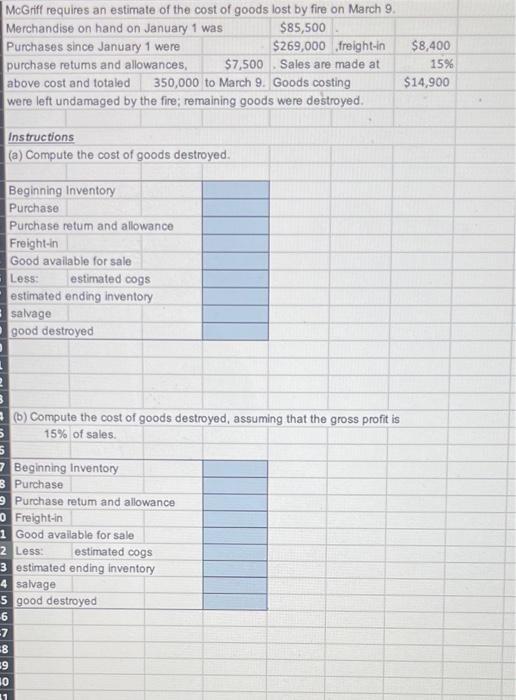
McGriff requires an estimate of the cost of goods lost by fire on March 9. $85,500...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
McGriff requires an estimate of the cost of goods lost by fire on March 9. $85,500 $269,000 freight-in Sales are made at Merchandise on hand on January 1 was Purchases since January 1 were purchase retums and allowances, above cost and totaled $7,500 350,000 to March 9. Goods costing were left undamaged by the fire; remaining goods were destroyed. 5 Instructions (a) Compute the cost of goods destroyed. -7 Beginning Inventory Purchase Purchase retum and allowance Freight-in Good available for sale Less: 3 Purchase 8 11 estimated cogs estimated ending inventory salvage good destroyed Purchase retum and allowance 0 Freight-in 1 Good available for sale 2 Less: estimated cogs 3 estimated ending inventory 4 salvage 5 good destroyed 6 39 10 (b) Compute the cost of goods destroyed, assuming that the gross profit is 15% of sales. Beginning Inventory $8,400 15% $14,900 McGriff requires an estimate of the cost of goods lost by fire on March 9. $85,500 $269,000 freight-in Sales are made at Merchandise on hand on January 1 was Purchases since January 1 were purchase retums and allowances, above cost and totaled $7,500 350,000 to March 9. Goods costing were left undamaged by the fire; remaining goods were destroyed. 5 Instructions (a) Compute the cost of goods destroyed. -7 Beginning Inventory Purchase Purchase retum and allowance Freight-in Good available for sale Less: 3 Purchase 8 11 estimated cogs estimated ending inventory salvage good destroyed Purchase retum and allowance 0 Freight-in 1 Good available for sale 2 Less: estimated cogs 3 estimated ending inventory 4 salvage 5 good destroyed 6 39 10 (b) Compute the cost of goods destroyed, assuming that the gross profit is 15% of sales. Beginning Inventory $8,400 15% $14,900
Expert Answer:
Answer rating: 100% (QA)
a gross profit so sales 100 cost 15 profit sales gross profit then cost of good ... View the full answer

Related Book For 

Intermediate Accounting
ISBN: 978-1118147290
15th edition
Authors: Donald E. Kieso, Jerry J. Weygandt, and Terry D. Warfield
Posted Date:
Students also viewed these accounting questions
-
McGriff requires an estimate of the cost of goods lost by fire on March 9. Merchandise on hand on January 1 was $76,000. Purchases since January 1 were $144,000; freight-in, $6,800; purchase returns...
-
Gross Profit Method Zidek Corp. requires an estimate of the cost of goods lost by fire on March 9. Merchandise on hand on January 1 was $38,000. Purchases since January 1 were $92,000; freight-in,...
-
Selected data from a February payroll register for Halverson Company are presented below. Some amounts are intentionally omitted. FICA taxes are 7.65%. State income taxes are 4% of gross earnings....
-
Tomas needs to borrow $3900 so he can repair damages to his car. He plans to pay the loan back in full in 5 months using his income tax refund. He can go to a finance company that will charge him 18%...
-
Greenwood Company manufactures two products-14,000 units of Product Y and 6,000 units of Product Z. The company uses a plantwide overhead rate based on direct labor-hours. It is considering...
-
Determining the limitations of the constitutional doctrine of due process has been a difficult issue to resolve. Use the Internet to research specific California cases in which a court considered...
-
China Express owns a number of stores that sell fast food. As part of its compensation packages, China Express provides employees with bonuses based on customer satisfaction surveys. Recent analysis...
-
Obtain the technology matrix from the input-output table. to A B C from A 0 200 300 B 500 400 300 C 0 0 600 Total Output 1,000 2,000 3,000
-
1. What type of attitude scale is used by the ACSI if any? 2. What type of attitude study might you design if you were the consultant? 3. How would you use results from this approach to diagnose...
-
A and B students are vying for the final battle in the National IE Strategy game. A students have formulated four strategies on how to win over B students while B students, being confident, only...
-
Write about NCell Based on the data for Equity Valuation Financial Highlights ( FY 2 0 2 1 / 2 2 ) : 1 Revenue NPR 3 2 . 2 4 billion 2 Profit before tax NPR 1 3 . 1 5 billion 3 Earnings per share (...
-
A parent acquires all of the outstanding common stock of a subsidiary for a cash purchase price of $ 1 , 6 0 0 , 0 0 0 . One reason the parent agreed to pay more than book value for the subsidiary is...
-
A certain fuse "blows" if the current in it exceeds 1.3 A, at which instant the fuse melts with a current density of 740 A/cm. What is the diameter of the wire in the fuse (in mm)? Your Answer:
-
How do different substances ( lotion, water, hand sanitizer and hair gel ) affect the behavior of electrostatically charged objects? Explain.
-
assume Noah wants to earn a return of 1 5 . 7 5 % and is offered the opportunity to purchase a $ 1 , 0 0 0 par value bond that pays a 1 3 . 5 0 % coupon rate ( distributed semiannually ) with three...
-
write the SQL code for these queries 2. Provide information about each fitness instructor's class schedule, including the instructor's name and id, information about the class itself, the number of...
-
Draw two scatterplots, one for which r = 1 and a second for which r = 21.
-
Southeast Airlines Inc. awards members of its Flightline program a second ticket at half price, valid for 2 years anywhere on its flight system, when a full-price ticket is purchased. How would you...
-
Nottebart Corporation has outstanding 10,000 shares of $100 par value, 6% preferred stock and 60,000 shares of $10 par value common stock. The preferred stock was issued in January 2014, and no...
-
(Need for GAAP ) Some argue that having various organizations establish accounting principles is wasteful and inefficient. Rather than mandating accounting rules, each company could voluntarily...
-
Clausius-Clayperon equation is applicable to (a) Solid-vapour (b) Solid-liquid (c) Liquid-vapour (d) All of these.
-
The reaction \(A(\mathrm{l}) ightarrow R(\mathrm{~g})\) is allowed to reach equilibrium condition in an autoclave. At equilibrium there are two phases - one a pure liquid phase of \(A\) and the other...
-
The equilibrium constant for the reaction \(\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}\) is 0.1084 . Under the same conditions, the equilibrium constant for...

Study smarter with the SolutionInn App


