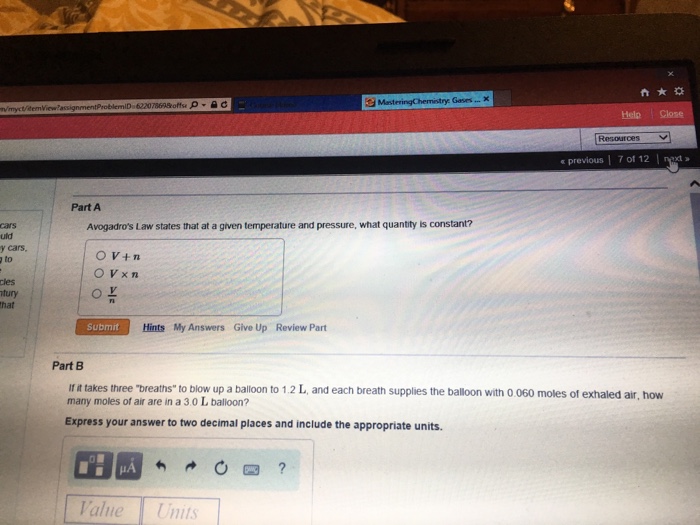
m/myct/itemView/assignment ProblemiD-62207869&offs D-AC cars uld y cars, to - cles tury that Part A Avogadro's Law...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
m/myct/itemView/assignment ProblemiD-62207869&offs D-AC cars uld y cars, to - cles tury that Part A Avogadro's Law states that at a given temperature and pressure, what quantity is constant? OV+n OVXn Submit HA Hints My Answers Give Up Review Part Value MasteringChemistry Gases... X Units ? Part B If it takes three "breaths" to blow up a balloon to 1.2 L, and each breath supplies the balloon with 0.060 moles of exhaled air, how many moles of air are in a 3.0 L balloon? Express your answer to two decimal places and include the appropriate units. 門 大亞 Help Close Resources < previous 7 of 12 next > m/myct/itemView/assignment ProblemiD-62207869&offs D-AC cars uld y cars, to - cles tury that Part A Avogadro's Law states that at a given temperature and pressure, what quantity is constant? OV+n OVXn Submit HA Hints My Answers Give Up Review Part Value MasteringChemistry Gases... X Units ? Part B If it takes three "breaths" to blow up a balloon to 1.2 L, and each breath supplies the balloon with 0.060 moles of exhaled air, how many moles of air are in a 3.0 L balloon? Express your answer to two decimal places and include the appropriate units. 門 大亞 Help Close Resources < previous 7 of 12 next >
Expert Answer:
Answer rating: 100% (QA)
Part A Avogadros Law states that at a given temperature and pre... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
How many protons and neutrons are in each isotope? c. 56Fe d. 224Rn EAl
-
How many protons and neutrons are in each isotope? c. 252Es d. 40K 48
-
How many millimeters of mercury are in 0.334 atm?
-
Consider the problem of minimizing (x, y) = x subject to g(x, y) = (x 1) 3 y 2 = 0. (a) Show, without using calculus, that the minimum occurs at P = (1, 0). (b) Show that the Lagrange condition P =...
-
You are conducting a market research project for FUN Inc. Given the project network that follows, complete the forward and backward pass, compute activity slack, and identify the critical path. Use...
-
John Murley operates John?s Cleaning Service. As the bookkeeper, you have been requested to journalize the following transactions: The chart of accounts for John?s Cleaning Service is as follows:...
-
What documents should you attempt to obtain before a lawsuit is filed during the informal fact-gathering stage? What sources are available to you to locate these documents?
-
It seems as if consolidated net income is always less than the sum of the parent's and subsidiary's separately calculated net incomes. Is it possible that the consolidated net income of the two...
-
Information, data, information, and insight are firmly related ideas, yet each plays its part disturbing the other, and each term has its significance. As per a typical view, information are gathered...
-
John Parsons (123-45-6781) and George Smith (123-45-6782) are 70% and 30% owners, respectively, of Premium, Inc. (11-1111111), a candy company located at 1005 16th Street, Cut and Shoot, TX 77303....
-
A company is setting its direct materials and direct labor standards for its leading product. Direct materials cost from the supplier are $5.00 per square foot, net of purchase discount. Freight-in...
-
If \(X_{t} \sim N(0, t)\), calculate the distribution of \(\left|X_{t} ight|\). Calculate \(\mathbf{E}\left|X_{t} ight|\) and \(V\left(\left|X_{t} ight| ight)\).
-
You are faced with making a decision on a large capital investment proposal. The capital investment amount is $\$ 640,000$. Estimated annual revenue at the end of each year in the eight-year study...
-
Consider the following normal mixture density: \[f(x)=0.7 \frac{1}{\sqrt{2 \pi 9}} e^{-\frac{(x-2)^{2}}{18}}+0.3 \frac{1}{\sqrt{2 \pi 4}} e^{-\frac{(x+1)^{2}}{8}}\]
-
If \(X_{t} \sim N\left(0, \sigma^{2} t ight)\) and \(Y_{t}=e^{X_{t}}\), calculate the pdf of \(Y_{t}\). Calculate \(\mathbf{E}\left[Y_{t} ight]\) and \(V\left(Y_{t} ight)\). Calculate the transition...
-
A piece of electronic circuitry embedded in a layer of diamond \(\left(n_{\text {diamond }}=2.42 ight.\) ) emits light. The diamond is covered by a coating of flint glass \(\left(n_{\text {flint...
-
Describe and explain how mindfulness works, and compare it to meditation. What are the stages of sleep, and how can you improve your sleep? explain
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Acetic acid can be manufactured by combining methanol with carbon monoxide, an example of a carbonylation reaction: (a) Calculate the equilibrium constant for the reaction at 25°C. (b)...
-
(a) If HNO3 is added to water, how does [OH-] change? How does pH change?
-
Which type or types of nuclear reactors have these characteristics? (a) Can use natural uranium as a fuel (b) Does not use a moderator (c) Can be refueled without shutting down
-
Derive the equation of motion and natural frequency for mass \(m\) shown in Figure 2.57. Assume the pulleys are massless, the cable is inextensible, and let the spring constant be \(k\). eeeee k...
-
Derive the equation of motion and natural frequency for mass \(m\) shown in Figure 2.58. Assume the pulleys are massless, and let the spring constants be \(k_{1}\) and \(k_{2}\). Let \(x_{1}\) and...
-
Show that the period of free vibration of a load weighing \(W\) suspended from two parallel springs, as shown in Figure 2.59, is given by \(T\), \[ T=2 \pi \sqrt{\frac{W}{g\left(k_{1}+k_{2} ight)}}...

Study smarter with the SolutionInn App


