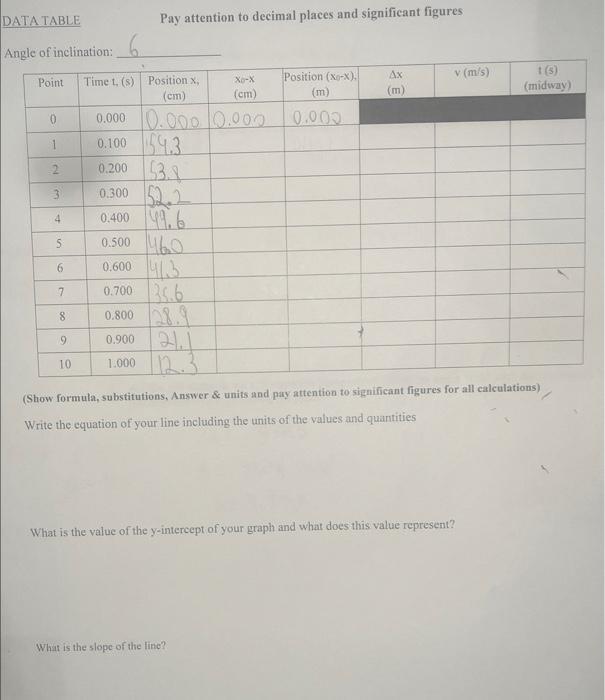
DATA TABLE Angle of inclination: Point 0 1 2 3 4 5 6 7 8 9...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
DATA TABLE Angle of inclination: Point 0 1 2 3 4 5 6 7 8 9 10 Pay attention to decimal places and significant figures 6 Time t, (s) Position X, (cm) 0.000 0.000 0.000 0.100 4.3 0.2003. 0.300 22 0.400 46 0.500 460 0.600 4 0.700 0.800 0.900 1.000 35.6 Xo-X (cm) Position (X-X). (m) 0.000 What is the slope of the line?? Ax v (m/s) (Show formula, substitutions, Answer & units and pay attention to significant figures for all calculations) Write the equation of your line including the units of the values and quantities What is the value of the y-intercept of your graph and what does this value represent? t(s) (midway) DATA TABLE Angle of inclination: Point 0 1 2 3 4 5 6 7 8 9 10 Pay attention to decimal places and significant figures 6 Time t, (s) Position X, (cm) 0.000 0.000 0.000 0.100 4.3 0.2003. 0.300 22 0.400 46 0.500 460 0.600 4 0.700 0.800 0.900 1.000 35.6 Xo-X (cm) Position (X-X). (m) 0.000 What is the slope of the line?? Ax v (m/s) (Show formula, substitutions, Answer & units and pay attention to significant figures for all calculations) Write the equation of your line including the units of the values and quantities What is the value of the y-intercept of your graph and what does this value represent? t(s) (midway)
Expert Answer:
Answer rating: 100% (QA)
position cm 50 45 40 35 30 25 20 15 10 5 0 0 02 04 X Here y is in cm X is in cm Else is constant Li... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these physics questions
-
A heat engine whose thermal efficiency is 35 percent uses a hot reservoir at 1100 R and a cold reservoir at 550 R. Calculate the entropy change of the two reservoirs when 1 Btu of heat is transferred...
-
A heat engine exhausts its heat a t35oC and has a Carnot efficiency of 39%. What exhaust temperature would enable it to achieve a Carnot efficiency of 49%?
-
A heat engine operates at a thermal efficiency that is 45% of the Carnot efficiency. If the temperatures of the high-temperature and low-temperature reservoirs are 400 oC and 50 oC, respectively,...
-
Starting with acetylene as your only source of carbon atoms, identify how you would prepare each member of the following homologous series of aldehydes: a. Ethanal b. Propanal c. Butanal d. Pentanal
-
For high school students graduating in 2013, college admissions to the nation's most selective schools were the most competitive in memory. Harvard accepted about 5.8% of its applicants, Dartmouth...
-
Use mathematical software to estimate the -electron binding energy of (a) Anthracene (8), (b) Phenanthrene (9) within the Huckel approximation.
-
An incompressible fluid oscillates harmonically \(\left(V=V_{0} ight.\) \(\sin \omega t\), where \(V\) is the velocity) with a frequency of \(10 \mathrm{rad} / \mathrm{s}\) in a 4-in.-diameter pipe....
-
What is a pass-through agency fund and under what conditions is it appropriate to use such a fund?
-
Explain the interplay between thermodynamics and statistical mechanics in describing the behavior of large ensembles of particles, focusing on the connection between macroscopic thermodynamic...
-
Background: A new ownership group has recently purchased ABC Liquors. You have been hired by the new management team to analyze their sales data for the past year and provide them with insights about...
-
choose the ratios that are industry specific measures of performance and be able to explain why those are the superior ratios by which to judge performance in that specific industry. Provide sample...
-
strictly evaluate the questions as you are thinking about them in general, NOT in relation to the product. i DO NOT want to hear that they need a good streaming TV service. I want to hear about their...
-
Drinder corp has declared that will offer dividend per share of 1.50 one year from now, 2 two year from now 1.75 three years from now you believe that you can sell the stock at the end of the third...
-
Consider a project with the following information: initial investment $750,000; straight-line depreciation to zero over five-year life; $25,000 salvage value; price per unit $76; variable cost per...
-
calculate the value of Brie using the capitalized EBITDA multiple approach b) calculate the value of Brie using the discounted cash flow approach Hint: estimate the next five years free cash flow...
-
The following data is given regarding three investments Gov Bond, Zam Beef and Zam Chicken. Predicted rate of return (%) State Probability GOV BOND ZAM BEEF ZAM CHICKEN Boom 0.3 10 25 14 Normal 0.45...
-
A borrower obtains an Interest-Only (IO) $2688 mortgage as a 30-year, monthly payment, fixed rate loan and with 7% interest. What is their regularly scheduled monthly payment?
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
What do you think of a diet program that allows for generous amounts of bread and rice provided that no butter or margarine is added?
-
A cooling tower with a cooling capacity of 30 tons (105 kW) is claimed to evaporate 4000 kg of water per day. Is this a reasonable claim?
-
Water is boiled at 1 atm pressure in a coffee maker equipped with an immersion-type electric heating element. The coffee maker initially contains 1 kg of water. Once boiling started, it is observed...
-
LDDS continued to publicly report increasing profits and sales in the financial statements, which allowed it to acquire more companies with no limit to the growth of its stock price. True/False
-
LDDS suffered from high fixed costs coupled with a lack of technical knowledge and expertise in optimizing the configuration of circuits, which resulted in high line costs. True/False
-
WorldCom overstated its earnings by improper accounting for multiple element contracts. True/False

Study smarter with the SolutionInn App


