Question: depicts an electron transport system (ETS) within a mitochondrial cristae fold from an animal cell's mitochondrion. The red arrows illustrate direction of proton (H*)
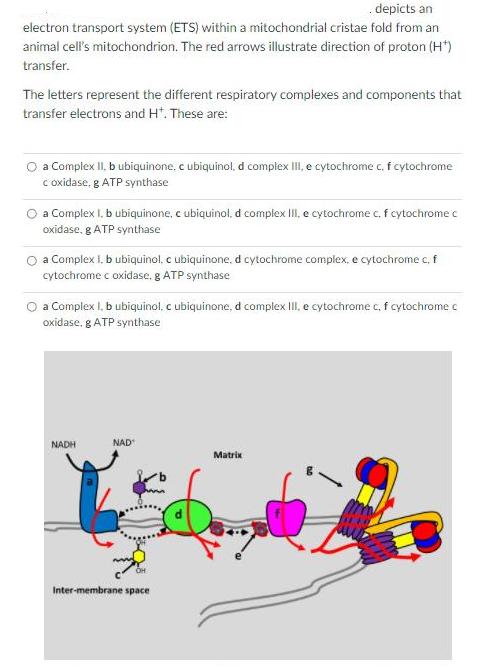
depicts an electron transport system (ETS) within a mitochondrial cristae fold from an animal cell's mitochondrion. The red arrows illustrate direction of proton (H*) transfer. The letters represent the different respiratory complexes and components that transfer electrons and H*. These are: O a Complex II, b ubiquinone, c ubiquinol, d complex III, e cytochrome c, f cytochrome c oxidase, g ATP synthase a Complex I. b ubiquinone, c ubiquinol, d complex II, e cytochrome c, f cytochrome c oxidase, g ATP synthase a Complex 1. b ubiquinol, c ubiquinone. d cytochrome complex, e cytochrome c, f cytochrome c oxidase, g ATP synthase a Complex I. b ubiquinol, c ubiquinone, d complex III, e cytochreme c, f cytochrome c oxidase, g ATP synthase NADH NAD Matrix Inter-membrane space
Step by Step Solution
3.40 Rating (159 Votes )
There are 3 Steps involved in it
Electron transport system within mitochondrial cristae In Electron t... View full answer

Get step-by-step solutions from verified subject matter experts


