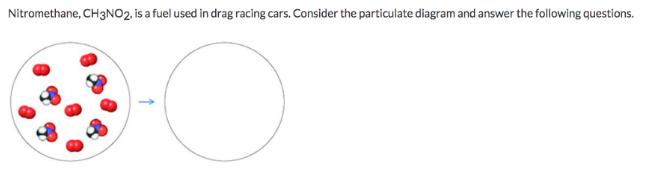
Nitromethane, CH3NO2, is a fuel used in drag racing cars. Consider the particulate diagram and answer...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Nitromethane, CH3NO2, is a fuel used in drag racing cars. Consider the particulate diagram and answer the following questions. Part 1 Write a balanced chemical equation describing the combustion of nitromethane.The nitrogen in the reactant is converted into N2(g) during the reaction. Part 2 Given the mixture reactants in the particulate model, how much of the excess reagent remains after the limiting reagent is completely consumed? If there is no limiting reagent (both reactants will be consumed equally), enter O in the space provided below. Nitromethane, CH3NO2, is a fuel used in drag racing cars. Consider the particulate diagram and answer the following questions. Part 1 Write a balanced chemical equation describing the combustion of nitromethane.The nitrogen in the reactant is converted into N2(g) during the reaction. Part 2 Given the mixture reactants in the particulate model, how much of the excess reagent remains after the limiting reagent is completely consumed? If there is no limiting reagent (both reactants will be consumed equally), enter O in the space provided below.
Expert Answer:
Answer rating: 100% (QA)
part 1 4CH 3 NO 2 l 3O 2 g 2N 2 g 4CO 2 g 6H 2 O g part 2 no of moles no of atoms Avogadro no ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Write a balanced chemical equation for each of the following reactions: (a) Phenol + sodium hydroxide (b) Product of part (a) + ethyl bromide (c) Product of part (a) + butyl p-toluenesulfonate (d)...
-
Write a balanced chemical equation using condensed structural formulas for the saponification (base hydrolysis) of (a) methyl propionate, (b) phenyl acetate.
-
Write a balanced chemical equation using condensed structural formulas for the saponification (base hydrolysis) of (a) methyl propionate, (b) phenyl acetate. Discuss.
-
4. Apple Inc. is a US-based technology company that plans to invest in a new research and development center in Europe and aims to raise the required funds as follows: < < - - A bond issue of...
-
Bunkhouse Electronics is a recently incorporated firm that makes electronic entertainment systems. Its earnings and dividends have been growing at a rate of 30%, and the current dividend yield is 2%....
-
In Fig. 29.8, if the angular speed v of the loop is doubled, then the frequency with which the induced current changes direction doubles, and the maximum emf also doubles. Why? Does the torque...
-
After the positrons were annihilated, the energy density of the universe was dominated by the photons and the neutrinos. Show that the energy density in that era was given by \(u_{\text {total...
-
On May 1, 2014, Leon Stoker opened Stokers Repair Service. During the month, he completed the following transactions for the company: May 1 Began business by depositing $10,000 in a bank account in...
-
3. (5 Marks) An engineering project requires $25,000 as first cost and has a planning horizon of six years. Operating costs are $2,300 per year, and expected annual revenue is $9,200 (both real...
-
Using Stellarium Web, change your location to the town of Auvers-sur-Oise by pasting 49.070720, 2.169886 into the location search box. Next, change your observing date to 1890-06-16. Look to the West...
-
Write an Executive Summary in which you analyze the liquidity, solvency and profitability of Wahlberg Company. Recommend with supporting points, whether the Company should be considered as an...
-
Research a company that has successfully integrated Six Sigma into its operational plan.Discuss how the company integrated and uses the principles.Give your opinion on its effectiveness within the...
-
Airbnb's vision is "to created a world where anyone can belong anywhere" and their mission is to "focus on creating an end-to-end travel platform that will handle every part of trip." Imagine you...
-
Is Elizabeth Holmes an overall ethical or unethical leader? From the podcast, readings and/or knowledge of the Holmes/Theranos situation answer this question by providing the following: 1. Ten clear...
-
Discuss the differences and relationshipsbetween a business-level strategy and a corporate-level strategy. Use examples from a company you are familiar with to illustrate these differencesand...
-
1. Do you support or oppose the United States' legal drinking age? Explain your stance. 2. Are there any circumstances under which adults should allow those under age 21 to consume alcohol? Identify...
-
How did Encyclopedia Britannica, Inc adapt its core business model to the information age? Did they succeed? Has Encyclopedia Britannica, Inc implemented a business or a digital transformation...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
The coordination number for the AI3+ ion is typically between four and six. Use the anion coordination number to determine the coordination number in the following compounds: (a) AlF3 where the...
-
Write balanced chemical equations to represent the following observations. (In some instances the complex involved has been discussed previously in the text.) (a) Solid silver chloride dissolves in...
-
How did Rutherford interpret the following observations made during his -particle scattering experiments? (a) Most particles were not appreciably deflected as they passed through the gold foil. (b)...
-
Explain the relationship between the finite sample \(F\)-test and the large sample \(\chi^{2}\)-test, and the assumptions under which each is suitable.
-
Use the Akaike information or Schwartz criteria to select variables for a predictive model.
-
Identify influential observations in a multiple regression model.

Study smarter with the SolutionInn App


