Question: 1. Kinetic energy of electron gas. Show that the kinetic energy of a three-dimensional gas of N free electrons at 0 K is U,
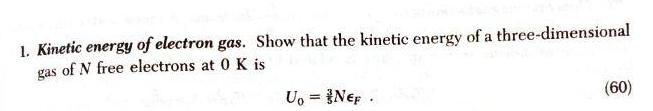
1. Kinetic energy of electron gas. Show that the kinetic energy of a three-dimensional gas of N free electrons at 0 K is U, = {Nep . (60)
Step by Step Solution
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Solution Accooding to feomi Disac Distoibution at OK we have F 1 ELEF ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


