6-30 Carbon dioxide at an initial pressure of 100 atm and a temperature of 300 K...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
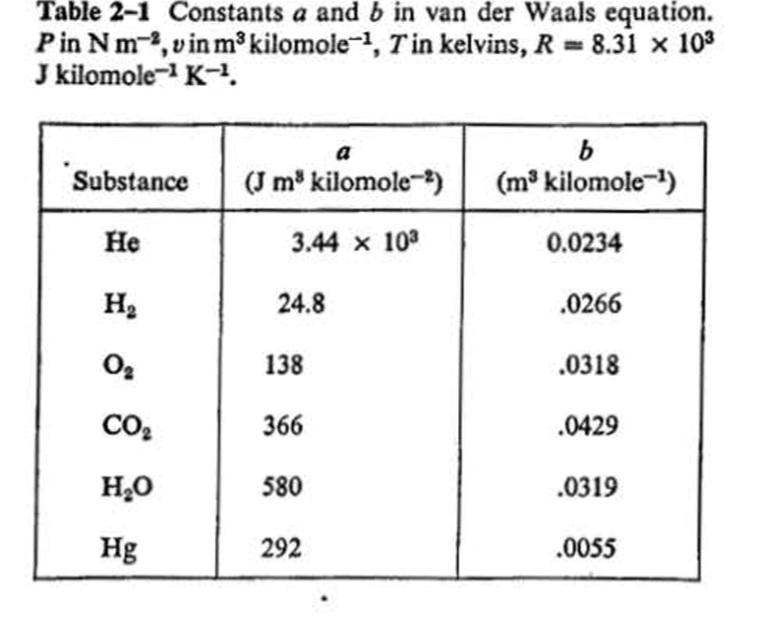
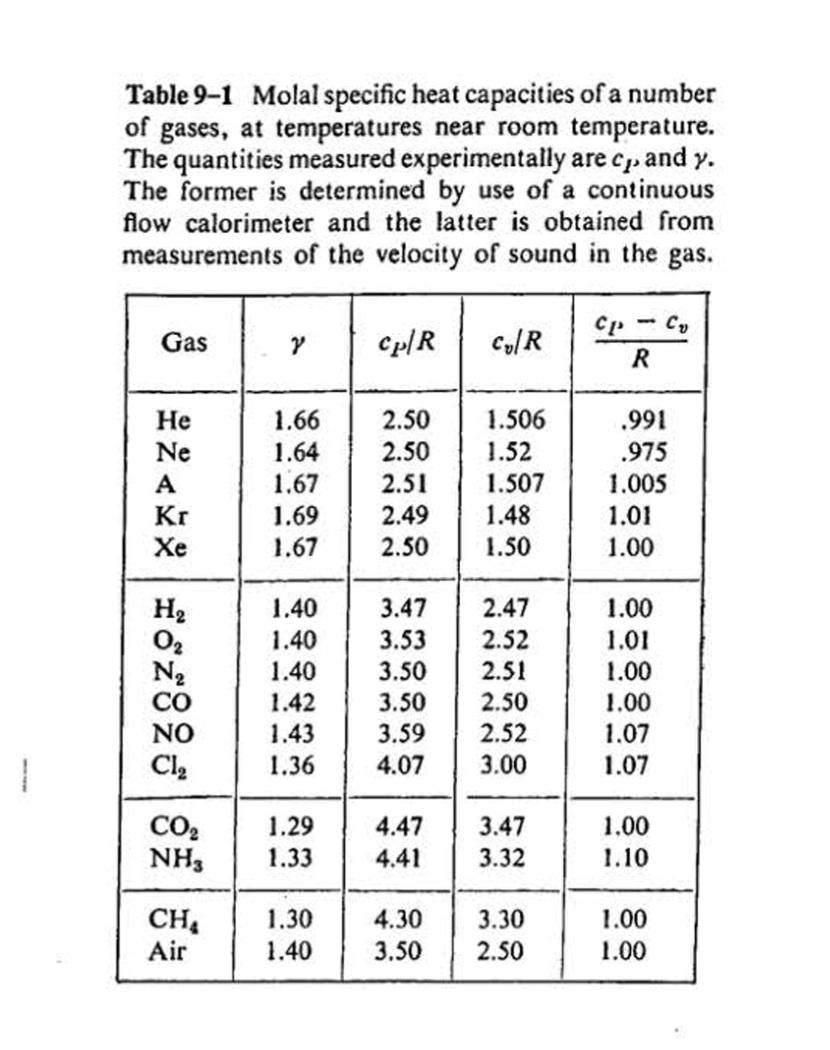
6-30 Carbon dioxide at an initial pressure of 100 atm and a temperature of 300 K under- goes an adiabatic free expansion in which the final volume is ten times the original volume. Find the change in temperature and the increase in specific entropy, assuming that CO, is (a) an ideal gas, (b) a van der Waals gas. (Use Tables 2-1 and 9-1 and make assumptions that seem reasonable.) any other Table 2-1 Constants a and b in van der Waals equation. Pin Nm,vin m³ kilomole-, Tin kelvins, R = 8.31 x 103 J kilomole- K-, a Substance (J m kilomole-) (m³ kilomole-) Не 3.44 x 10 0.0234 H2 24.8 .0266 138 .0318 CO2 366 .0429 H,0 580 .0319 Hg 292 .0055 Table 9-1 Molal specific heat capacities of a number of gases, at temperatures near room temperature. The quantities measured experimentally are cp. and y. The former is determined by use of a continuous flow calorimeter and the latter is obtained from measurements of the velocity of sound in the gas. Cp-C, Gas Cp/R Cy/R Не 1.66 2.50 1.506 .991 2.50 1.52 1.507 Ne 1.64 .975 A 1.67 2.51 1.005 Kr 1.69 2.49 1.48 1.01 Хе 1.67 2.50 1.50 1.00 H2 1.40 3.47 2.47 1.00 1.40 3.53 2.52 1.01 N2 CO 1.40 3.50 2.51 1.00 1.42 1.43 3.50 2.50 1.00 NO 3.59 2.52 1.07 Cl, 1.36 3.00 1.07 CO2 NH, 1.29 4.47 3.47 1.00 .33 4.41 3.32 1.10 CH Air 1.30 4.30 3.30 1.00 1.40 3.50 2.50 1.00 6-30 Carbon dioxide at an initial pressure of 100 atm and a temperature of 300 K under- goes an adiabatic free expansion in which the final volume is ten times the original volume. Find the change in temperature and the increase in specific entropy, assuming that CO, is (a) an ideal gas, (b) a van der Waals gas. (Use Tables 2-1 and 9-1 and make assumptions that seem reasonable.) any other Table 2-1 Constants a and b in van der Waals equation. Pin Nm,vin m³ kilomole-, Tin kelvins, R = 8.31 x 103 J kilomole- K-, a Substance (J m kilomole-) (m³ kilomole-) Не 3.44 x 10 0.0234 H2 24.8 .0266 138 .0318 CO2 366 .0429 H,0 580 .0319 Hg 292 .0055 Table 9-1 Molal specific heat capacities of a number of gases, at temperatures near room temperature. The quantities measured experimentally are cp. and y. The former is determined by use of a continuous flow calorimeter and the latter is obtained from measurements of the velocity of sound in the gas. Cp-C, Gas Cp/R Cy/R Не 1.66 2.50 1.506 .991 2.50 1.52 1.507 Ne 1.64 .975 A 1.67 2.51 1.005 Kr 1.69 2.49 1.48 1.01 Хе 1.67 2.50 1.50 1.00 H2 1.40 3.47 2.47 1.00 1.40 3.53 2.52 1.01 N2 CO 1.40 3.50 2.51 1.00 1.42 1.43 3.50 2.50 1.00 NO 3.59 2.52 1.07 Cl, 1.36 3.00 1.07 CO2 NH, 1.29 4.47 3.47 1.00 .33 4.41 3.32 1.10 CH Air 1.30 4.30 3.30 1.00 1.40 3.50 2.50 1.00
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
1 (one) kg of air at an initial pressure of 8 bar and 100 C is expanded polytropically with n = 1,2 so that the pressure drops as much as one-fifth of the initial pressure. Determine: a. Specific...
-
A gas has an initial pressure of 1.445 atm and an initial volume of 1.009 L. What is its new pressure if volume is changed to 0.556 L? Assume temperature and amount are held constant.
-
A gas has an initial pressure of 4.33 atm and an initial volume of 5.88 L. What is its new volume if pressure is changed to 0.506 atm? Assume temperature and amount are held constant.
-
According to Michel Foucault, while it is an advancement to no longer torture persons, modern forms of disciplinary punishment are sinister because they strive for _____________. a. routines. b....
-
Suppose that people expect deflation (a falling price level), but aggregate demand remains at AD0. a. What happens to the short-run and long-run aggregate supply curves? (Draw some new curves if you...
-
The Classic Theater is owned by Kim Lockerby. All facilities were completed on March 31. At this time, the ledger showed: No. 101 Cash $4,000, No. 140 Land $10,000, No. 145 Buildings (concession...
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Sandi Scott obtained a patent on a small electronic device and organized Scott Products, Inc., to produce and sell the device. During the first month of operations, the device was very well received...
-
ony purchased an empty office building in London from Margot, an unconnected third party, on 1 September 2024. Tony paid 996,000 for the office building. No VAT was charged on the supply. Margot had...
-
Lisbon Company SA lost 70% of its inventory in a fire on March 25, 2017. The accounting records showed the following gross profit data for February and March. Lisbon Company is fully insured for fire...
-
Jordan Peters works for Protea Ltd and is paid $2,250 per week. He is entitled to two week's vesting sick leave per annum. During May 2021, Jordan was off work on sick leave for the whole week...
-
The financial manager of Solsken is evaluating a proposal to purchase a new solar machine unit that has a lifetime of 10 years. The new machine would allow the company to make cost savings of SKr4...
-
In the previous problem, suppose the project requires an initial investment in net working capital of 2,000 and the investment will have a market value of 1,000 at the end of the project. Assume the...
-
Car manufacturers BMW have made extensive use of temporary labour since they began production of the Mini at their Oxford plant in the UK in 2001. Entering a different market segment from their...
-
Evaluation of performance, merit-rating, or annual review. It nourishes short-term performance, annihilates long-term planning, builds fear, demolishes teamwork, and nourishes rivalry and politics....
-
Cultural approaches to comparing HR between contexts tend to assume that countries do not readily change their defining cultural identities, and this will exert long-term effects on work and...
-
A fire destroyed equipment used by Pirates Inc. in its manufacturing business. Pirates' adjusted tax basis in the equipment was $24,000 Three weeks after the fire, Pirates paid $40,000 for a...
-
Could the owner of a business prepare a statement of financial position on 9 December or 23 June or today?
-
A rifle bullet with a muzzle speed of 330 m/s is fired directly into a special dense material that stops the bullet in 25.0 cm. Assuming the bullets deceleration to be constant, what is its magnitude?
-
Find the currents in the circuit branches in Fig 18.38. 4.0 R5-2.0 R: 2.0 Re 2.0 5.0 6.0
-
The following is an old trick (Fig. 4.28). If a tablecloth is yanked out very quickly, the dishes on it will barely move. Why?
-
Determine the equivalent resistance \(R_{\mathrm{eq}}\) for the circuit shown in Figure 6.11. Assume that all resistors have the same resistance of \(R\). FIGURE 6.11 Problem 4. R W R3 BA w R ww
-
Determine the equivalent resistance \(R_{\text {eq }}\) for the circuit shown in Figure 6.10. Assume that all resistors have the same resistance of \(R\). FIGURE 6.10 Problem 3. W R . WWW R5 w R2 R3...
-
The output voltage of a voltage divider is not fixed; it varies according to the load. a. Find the output voltage \(v_{\mathrm{o}}\) in Figure 6.13 for two different values of load resistance: (1)...

Study smarter with the SolutionInn App


