6-6 A substance has the properties that (au/dv)T the equation of state must be T APv...
Fantastic news! We've Found the answer you've been seeking!
Question:
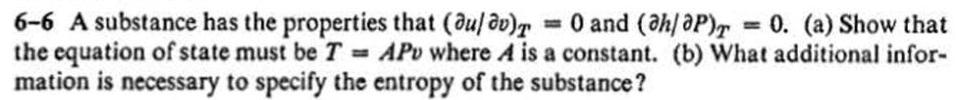
Transcribed Image Text:
6-6 A substance has the properties that (au/dv)T the equation of state must be T APv where A is a constant. (b) What additional infor- mation is necessary to specify the entropy of the substance? = 0 and (ah/ aP)r = 0. (a) Show that %3D %3D %3D 6-6 A substance has the properties that (au/dv)T the equation of state must be T APv where A is a constant. (b) What additional infor- mation is necessary to specify the entropy of the substance? = 0 and (ah/ aP)r = 0. (a) Show that %3D %3D %3D
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these physics questions
-
Given the following information, prepare a statement of cash flows. Increase in accounts receivable $35 Increase in inventories 40 Operating income 95 Interest expense 35 Increase in accounts payable...
-
Please help me with this its urgent please help me for freee How will the social sciences affect your life in the following areas and explain why: Area of Life Effect and why Work / Career Home and...
-
Number 10.11, 10.12 . Please help me
-
The San Francisco Chronicle reported that two Stanford graduates, Dave Kaval and Brad Null, set a goal to see a game in every major league baseball stadium. They began in San Francisco and selected...
-
Use a computer to demonstrate the truth of the theory presented in this section. a. The underlying assumptions are the populations are normally distributed, and while conducting a hypothesis test for...
-
Orange light of wavelength 0.61 m in air enters a block of glass with r = 1:44. What color would it appear to a sensor embedded in the glass? The wavelength ranges of colors are violet (0.39...
-
Widget, Corp., completed the following inventory transactions during the month of January: Requirements 1. Without resorting to calculations, determine which inventory method will result in Widget,...
-
A hydrogen atom in the 5g state is placed in a magnetic field of 0.600 T that is in the z-direction. (a) Into how many levels is this state split by the interaction of the atom's orbital magnetic...
-
Discuss the justification and how to measure the ROI of social commerce and the risk factors and implementation issues associated with social commerce and how to build a strategy for success in...
-
Consider a hospital in which each doctor is responsible for many patients while each patient is cared for by just one doctor. Each doctor has a unique employee number, name, telephone number, and...
-
Operating systems and application programs play vital role in our daily usage of computers. Differentiate between an operating system and an application program give examples each.
-
Aniyah earned $25,000 as an early childhood teacher in the Lindbergh School District. She was promoted to a sixth grade teaching position that included an 80% raise. What will she earn as a new sixth...
-
Examine the areas of biases in organizational decision making and within the selection process for a diverse workforce. Discuss two ways to overcome these biases.
-
Arman Corporation's relevant range of activity is 4,200 units to 9,800 units. When it produces and sells 7,000 units, its average costs per unit are as follows: Average Cost per Unit Direct labor...
-
How many kanban cards are necessary for a process that uses 230 units per shift, the lead time for the containers is 4 shifts and a safety stock of 25 units. Each container manage 48 units.
-
Use the CAPM to find the required return for asset X and Y. Asset X: Year 2013 2014 2015 2016 2017 2018 2019 2020 2021 2022 Cash Flow $1,000 $1,500 $1,400 $1,700 $1,900 $1,600 $1,700 $2,000 $2,100...
-
Suppose a company has proposed a new 4-year project. The project has an initial outlay of $59,000 and has expected cash flows of $19,000 in year 1, $23,000 in year 2, $27,000 in year 3, and $46,000...
-
Air pollution generated by a steel mill is an example of a) a positive production externality. b) a negative production externality. c) a public good. d) the free-rider problem. State and local taxes...
-
(a) Increase [NH3], increase yield NO (b) Increase [H2O], decrease yield NO (c) Decrease [O2], decrease yield NO (d) Decrease container volume, decrease yield NO (fewer moles gas in reactants) (e)...
-
The average concentration of carbon monoxide in air in an Ohio city in 2006 was 3.5 ppm. Calculate the number of CO molecules in 1.0 L of this air at a pressure of 759 torr and a temperature of 22oC.
-
Two compounds have the same empirical formula. One substance is a gas, whereas the other is a viscous liquid. How is it possible for two substances with the same empirical formula to have markedly...
-
Use Rayleigh's method to solve Problem 2.26. Data From Problem 2.26:- A mass \(m\) is attached to a cord that is under a tension \(T\), as shown in Fig. 2.67. Assuming that \(T\) remains unchanged...
-
Use Rayleigh's method to solve Problem 2.96. Data From Problem 2.96:- Find the equation of motion of the uniform rigid bar \(O A\) of length \(l\) and mass \(m\) shown in Fig. 2.104. Also find its...
-
Use the energy method to find the natural frequency of the system shown in Fig. 2.103. Figure 2.103:- k m, Jo k 000 R FIGURE 2.103 Cylinder restrained by springs.

Study smarter with the SolutionInn App



