? If 4.0 g of acetanilide are mixed with 1.5 mL of 16M nitric acid and excess
Fantastic news! We've Found the answer you've been seeking!
Question:
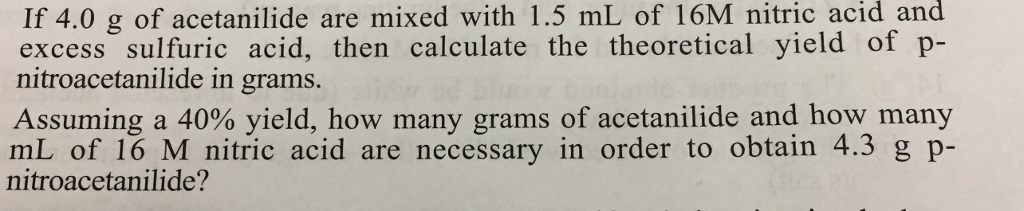
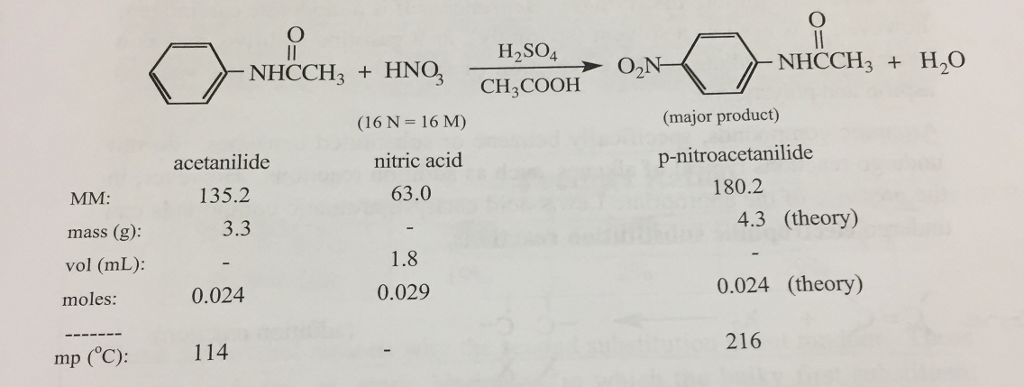
Transcribed Image Text:
If 4.0 g of acetanilide are mixed with 1.5 mL of 16M nitric acid and excess sulfuric acid, then calculate the theoretical yield of p- nitroacetanilide in grams. Assuming a 40% yield, how many grams of acetanilide and how many mL of 16 M nitric acid are necessary in order to obtain 4.3 g p- nitroacetanilide? MM: mass (g): vol (ml): moles: mp (°C): acetanilide 135.2 3.3 0.024 NHCCH3 + HNO3 114 (16 N = 16 M) nitric acid 63.0 - 1.8 0.029 H₂SO4 CH3COOH NHCCH3 + H,O (major product) p-nitroacetanilide 180.2 4.3 (theory) 0.024 (theory) 216 If 4.0 g of acetanilide are mixed with 1.5 mL of 16M nitric acid and excess sulfuric acid, then calculate the theoretical yield of p- nitroacetanilide in grams. Assuming a 40% yield, how many grams of acetanilide and how many mL of 16 M nitric acid are necessary in order to obtain 4.3 g p- nitroacetanilide? MM: mass (g): vol (ml): moles: mp (°C): acetanilide 135.2 3.3 0.024 NHCCH3 + HNO3 114 (16 N = 16 M) nitric acid 63.0 - 1.8 0.029 H₂SO4 CH3COOH NHCCH3 + H,O (major product) p-nitroacetanilide 180.2 4.3 (theory) 0.024 (theory) 216
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Two moles of H2 are mixed with 1 mol O2 at 25oC. No observable reaction takes place, although Kc for the reaction to form water is very large at this temperature. When a piece of platinum is added,...
-
Calculate the theoretical yield of 2,3-dibromo-3-phenylpropanoic acid starting with 2.56g of trans-cinnamic acid (TCA). a. What volume of 1.2M bromine in acetic acid is required to fully convert the...
-
Sulfuric acid (H2SO4), the most important industrial chemical in the world, is prepared by oxidizing sulfur to sulfur dioxide and then to sulfur trioxide. Although sulfur trioxide reacts with water...
-
A mixture of 1 kmol carbon dioxide, 2 kmol carbon monoxide, and 2 kmol oxygen, at 25C, 150 kPa, is heated in a constant pressure steady state process to 3000 K. Assuming that only these same...
-
An insulated rigid tank is divided into two equal parts by a partition. Initially, one part contains 3 kg of argon gas at 300 kPa and 70C, and the other side is evacuated. The partition is now...
-
How is the experience and expression of emotion different in different types of jobs? Compare, for example, emotion in the work of a minister, a store clerk, and a data processor. Are the emotional...
-
The \(45^{\circ}\) off-axis test shown in Figure 10.37 is conducted on a 10 in. \(\times 1\) in. \(\times 0.1\) in. \((254 \mathrm{~mm} \times 25.4 \mathrm{~mm} \times 2.54 \mathrm{~mm})\)...
-
You have been asked to advise a business-to-business manufacturing company how to detect fraudulent financial reporting. Management does not understand how early revenue recognition by backdating...
-
Calculate the cost of debt. Calculate the cost of equity. Calculate the weight of debt and equity. Calculate the WACC. Table 4 Stock Info Amount Bond Info Amount Shares Outstanding 100 Face value...
-
When an investor uses the equity method to account for investments in common stock, the investors share of cash dividends from the investee should be recorded as a. A deduction from the investors...
-
9 Please write the output value of the following code. #include int main (void) { } char cstring[] = "hello"; int i=0, len=0; while(cstring[i] != '\0') { len++; i++;| } for (i = len-1; i >= 0; i--)...
-
What is the imperative mood, and why is it preferred over the indicative mood?
-
Explain the difference between brainstorming and brainwriting.
-
What is the benefit of dovetailing sentences within a paragraph?
-
What employability skills are Canadian employers looking for in an employee?
-
Suppose for the previous Problem 3.21, that your CEO is highly risk averse and will not invest in any project with a payback longer than three years. Assuming a required rate of return of 15%, what...
-
What recent advancements in respiratory physiology research have shed light on novel therapeutic approaches for managing respiratory diseases and optimizing respiratory performance in both health and...
-
Determine by direct integration the values of x for the two volumes obtained by passing a vertical cutting plane through the given shape of Fig. 5.21. The cutting plane is parallel to the base of the...
-
A single bond is almost always a sigma bond, and a double bond is almost always made up of a sigma bond and a pi bond. There are very few exceptions to this rule. Show that the B2 and C2 molecules...
-
Compare the heat produced by the complete combustion of 1 mole of methane (CH4) with a mole of water gas (0.50 mole H2 and 0.50 mole CO) under the same conditions. On the basis of your answer, would...
-
Draw structures for (a) (HF)2 and (b) HF2-
-
Show that having the exact value for the estimated sensor bandwidth is not required for the experimental process to find KEst discussed in Section 10. 4.3.4. In Experiment 10E, corrupt the value of...
-
Provide the modeling equations for the following figure. Assume an analog PI controller (GC(s)), a single-pole low-pass filter with a break frequency of 200 Hz for the feedback filter (H(s)), and a...
-
What is the gain and phase of a single-pole low-pass filter (a) at its bandwidth and (b) at 1/10 its bandwidth? (c) Repeat for a two-pole low-pass filter with = 0.7

Study smarter with the SolutionInn App



