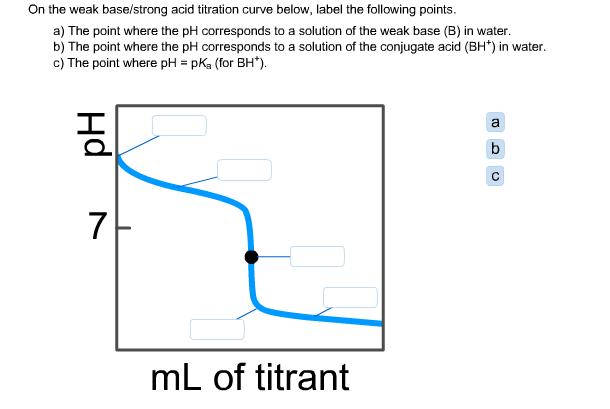
On the weak base/strong acid titration curve below, label the following points. a) The point where...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
On the weak base/strong acid titration curve below, label the following points. a) The point where the pH corresponds to a solution of the weak base (B) in water. b) The point where the pH corresponds to a solution of the conjugate acid (BH*) in water. c) The point where pH = pK₂ (for BH*). Hd 7 mL of titrant 10 D a b On the weak base/strong acid titration curve below, label the following points. a) The point where the pH corresponds to a solution of the weak base (B) in water. b) The point where the pH corresponds to a solution of the conjugate acid (BH*) in water. c) The point where pH = pK₂ (for BH*). Hd 7 mL of titrant 10 D a b
Expert Answer:
Answer rating: 100% (QA)
From given formula for T we I obtain T41 74 x X 7 4 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In general, as base strength increases, conjugate acid strength decreases. Explain why the conjugate acid of the weak base NH3 is a weak acid.
-
The figure compares the titration of a monoprotic weak acid with a monoprotic weak base and the titration of a diprotic acid with strong base. (a) Write the reaction between the weak acid and the...
-
A 0.01 M solution of a weak acid in water is 0.05% ionized at 25C. What is its pK?
-
Pacific Company sells electronic test equipment that it acquires from a foreign source. During the year 2014, the inventory records reflected the following: Inventory is valued at cost using the LIFO...
-
Is Marketing Management an Art or a Science? Some observers maintain that good marketing is mostly an art and does not lend itself to rigorous analysis and deliberation. Others contend it is a highly...
-
A classic example in coalitional game theory is the three-person majority game, in which the allocation of $1 among three persons is decided by majority vote. Let N = {1, 2, 3}. The characteristic...
-
Consider the explicit, fully implicit, and semi-implicit methods discussed in Section 10.3. Order them by the amount of computations required at every time step. Explain your answer.
-
The following data were taken from the financial statements of Ares Inc. for the current fiscal year. Assuming the long-term investments totaled $3,000,000 throughout the year and that total assets...
-
It is so hard to do it, I tried so many times Rocky Guide Service provides guided 175 day hiking tours throughout the Rocky Mountains. Wilderness Tours hires Rocky to lead van'ous tours that...
-
1. What is the cost of debt for Sunrise Bakery? 2. What is their cost of equity? 3. What is the WACC? 4. Which cost of capital should be used to evaluate the feasibility of the oven purchase? 5....
-
AT&T: Business acquisitions and analysis of sales growth (LO17-5) The disclosure rules for business combinations complicatefinancial analysis. Trend analysis becomes difficult becausecomparativ 2...
-
What is the required length of a straight and rigid beam that vibrates with an amplitude of 10 m in a plane perpendicular to the mean wind speed of 10 m/s if you assume that the power coefficient is...
-
A cooling crystallizer has a feed of 1,000 lb/h of 32.5 wt% MgSO4 in water at 120F. This solution is cooled to 70F to form crystals of the heptahydrate. The feed of the specific heat is approximately...
-
A grocery deliver service is guaranteeing an order fulfillment time of 4 hours (delivered to the customers doorstep) of the order is free. If the service fulfillment follows an exponential...
-
WHAT WAS THE JUDGMENT IN THE ARREST WARRANT CASE?
-
A review of accounting records for last year disclosed the following selected information: Variable costs: Direct materials used68,500 Direct labor177,500 Manufacturing overhead166,500 Selling...
-
Diameters data frame of the first sample (showing only the first five observations) diameters 0 1.99 1 1.56 2 1.89 3 2.74 4 1.81 Diameters data frame of the second sample (showing only the first five...
-
The first law of thermodynamics is sometimes whimsically stated as, You cant get something for nothing, and the second law as, You cant even break even. Explain how these statements could be...
-
The molar enthalpy of vaporization of water at 373 K is 41.16 kJ/mol. What fraction of this energy is used to change the internal energy of the water, and what fraction is used to do work against the...
-
Entropy can be calculated by a relationship proposed by Ludwig Boltzmann: S = kB ln V where kB = 1.38 Ã 1023 J/K and V is the number of ways a particular state can be obtained. (This equation...
-
At 1258C, Kp = 0.25 for the reaction 2NaHCO3(s) Na2CO3(s) + CO2(g) + H2O(g) A 1.00- L flask containing 10.0 g of NaHCO3 is evacuated and heated to 125oC. a. Calculate the partial pressures of CO2...
-
Where is the home office of the Association of Certified Fraud Examiners? Would all of its members have an accounting and auditing background? Does the group have a Code of Ethics?
-
Should a forensic accountant express an opinion in a report regarding the guilt or innocence of any person or party?
-
What certificate does the FASNA group issue?

Study smarter with the SolutionInn App


