One mole of solid silver superheated to 1000 C is allowed to melt at the same...
Fantastic news! We've Found the answer you've been seeking!
Question:
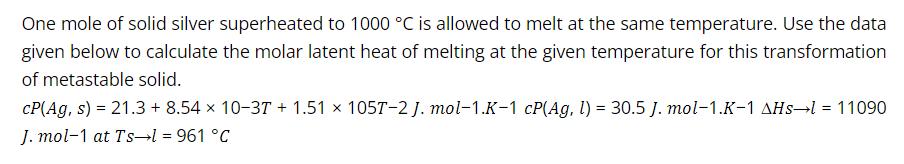
Transcribed Image Text:
One mole of solid silver superheated to 1000 °C is allowed to melt at the same temperature. Use the data given below to calculate the molar latent heat of melting at the given temperature for this transformation of metastable solid. CP(Ag, s) = 21.3 + 8.54 x 10-37 + 1.51 x 105T-2 J. mol-1.K-1 cP(Ag, l) = 30.5 J. mol-1.K-1 AHs-l = 11090 J. mol-1 at Ts l = 961 °C One mole of solid silver superheated to 1000 °C is allowed to melt at the same temperature. Use the data given below to calculate the molar latent heat of melting at the given temperature for this transformation of metastable solid. CP(Ag, s) = 21.3 + 8.54 x 10-37 + 1.51 x 105T-2 J. mol-1.K-1 cP(Ag, l) = 30.5 J. mol-1.K-1 AHs-l = 11090 J. mol-1 at Ts l = 961 °C
Expert Answer:
Answer rating: 100% (QA)
Date 1 20 Page No Guien Molei of Solid delver I mod Meltingpaint of Silber 961C Heat o... View the full answer

Posted Date:
Students also viewed these accounting questions
-
One mole of solid Cr 2 O 3 at 2500 K is dissolved in a large volume of a liquid Raoultian solution of Al 2 O 3 and Cr 2 O 3 in which X Cr2O3 = 0.2 and which is also at 2500 K. Calculate the changes...
-
One mole of solid gold is raised from 25 C to 100 C at constant pressure. Cp/(J/K mol) = 23.7 + 0.00519T. Calculate S for the transformation.
-
Two containers hold an ideal gas at the same temperature and pressure. Both containers hold the same type of gas, but container B has twice the volume of container A. (i) What is the average...
-
It is stated in Section 40.3 that a finite potential well always has at least one bound level, no matter how shallow the well. Does this mean that as U 0 0, E 1 0? Does this violate the Heisenberg...
-
1. If you were Beckwith, would you have dropped your transfer request to help Martin? 2. Should Fisher have given Martin preference over Beckwith because of his disability? Does seniority override...
-
1. What perspective of organizational effectiveness did Tina Stavros and James Alder attempt to apply in this case? Describe how specific elements of that perspective related to their interventions....
-
Two small, irregularly shaped conducting objects, one carrying charge \(+q\) and one carrying charge \(-q\), are placed on an \(x\) axis at \(x=-4.0 \mathrm{~m}\) and \(x=+4.0 \mathrm{~m}\),...
-
The plaintiff, Herbert Rosenthal Jewelry Corporation, and the defendant, Kalpakian, manufactured jewelry. The plaintiff obtained a copyright registration of a jeweled pin in the shape of a bee....
-
11. If dy + 2y tan x = sin x, 0
-
A wet t-shirt hung on a hanger has a total surface area of about 0.6 m2 . It loses water as follows: time (min) 0 5 33 45 weight (g) 661 640 580 553 Calculate the flux of water vapor away from the...
-
From the relational database schema 1. write sql query to display a list of available Drivers sorted according to their clearance level. Display the drive's license number, first name, last name and...
-
Penelope Morton's adjusted gross income (AGI) for 2022 was $157,000. She also received certain income benefits in 2022. She received $1,400 of state unemployment insurance benefits, $2,000 from a...
-
When developing proformas, it is common to add Reserves to the model. Reserves may represent a property owner's estimate of annual capital needs, or it may be the owner's view of the amount they need...
-
Talsa, Inc., an all-equity firm, operates in the growing electric vehicle market. Over the next three years (T=1,2,3), Talsa, Inc., is expected to generate FCF of $2.4 billion, $3.6 billion, and $5.0...
-
Under the terms of a partnership agreement, Erica is entitled to a fixed annual payment of $10,000 without regard to the income of the partnership. Her distributive share of the partnership income is...
-
The Kaur family want to visit a theme park while on holiday. The attraction has quoted you a net cost of $400.00 for four one-day entry passes. Your workplace's policy is to add a 15% markup on all...
-
What percentage of Canadians believe winning the lottery is the best path to financial success
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
Describe an example of resistance to change that you have observed. Why did it occur?
-
What qualities would the ideal gatekeeper possess to facilitate the communication of technical information in the firm?
-
Debate: Survey feedback can be a problematic OD technique because it permits people who are affected by organizational policies to generate data that speak against those policies.

Study smarter with the SolutionInn App

