Osmotic pressure of 30% solution of glucose is 1.20 atm and that of 3.42% solution of...
Fantastic news! We've Found the answer you've been seeking!
Question:
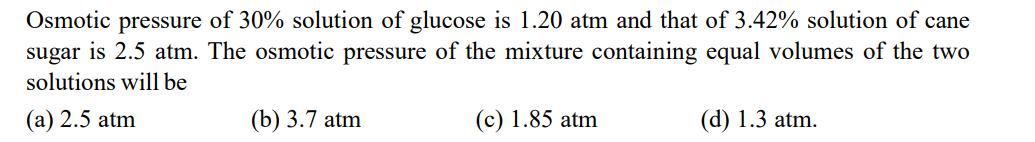
Transcribed Image Text:
Osmotic pressure of 30% solution of glucose is 1.20 atm and that of 3.42% solution of cane sugar is 2.5 atm. The osmotic pressure of the mixture containing equal volumes of the two solutions will be (a) 2.5 atm (b) 3.7 atm (c) 1.85 atm (d) 1.3 atm. Osmotic pressure of 30% solution of glucose is 1.20 atm and that of 3.42% solution of cane sugar is 2.5 atm. The osmotic pressure of the mixture containing equal volumes of the two solutions will be (a) 2.5 atm (b) 3.7 atm (c) 1.85 atm (d) 1.3 atm.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The essential structure of a certain type of aircraft turn indicator is shown. Each spring has a constant of 40 lb/ft, and the 7-oz uniform disk of 2-in. radius spins at the rate of 10 000 rpm. The...
-
Using the plurality with elimination method, which restaurant is chosen? The members of the Student Council at Ohio State University are planning to go out to dinner following an upcoming meeting....
-
Solutions A and B have osmotic pressures of 2.4 atm and 4.6 atm, respectively, at a certain temperature. What is the osmotic pressure of a solution prepared by mixing equal volumes of A and B at the...
-
The costs of achieving emission reductions in the future will depend greatly on the types of policies used to reduce emissions today. Explain.
-
During 2011, Rooster Company purchased 5,000 shares of Hen Company common stock for $18 per share and 3,200 shares of Egg Company common stock for $21 per share. These investments are intended to be...
-
Water accounts for 55% of a woman's weight (ga.water.usgs.gov/edu). Lily weighs 135 lb. How much of her body weight is water?
-
In Equation (3.153), we saw a linear version of an epidemic model. The commonly used nonlinear SIR model is given by \[\begin{align*} \frac{d S}{d t} & =-\beta S I \\ \frac{d I}{d t} & =\beta S...
-
Kathleen McFadden, vice president of operations at Jackson Manufacturing Company, has just received a request for quote (RFQ) from DeKalb Electric Supply for 400 units per week of a motor armature,...
-
Inventory Write-Down The following information for Tuell Company is available: Case 1 2 3 4 5 Cost Net realizable value $5.00 $5.00 $5.00 $5.00 $5.00 Net realizable value less normal profit...
-
Prepare the journal entries of the following transactions. 1. On January 1, 20XX, BVD Corp. issued 3,500 shares of $50 par, 7% preferred stock for $275,000. 2. On January 1, 20XX, BVD Corp. also...
-
1. Let W be the space spanned by f = sin x and g = cos x. a. Show that for any value of θ, f1 = sin(x+θ) and g1 =cos(x+θ) are vectors in W b. Show that f1 and g1 form a basis for W.
-
A is a U.S.-based MNC with AAA credit; B is an Italian firm with AAA credit. Firm A wants to borrow 1,000,000 for one year and B wants to borrow $2,000,000 for one year. The spot exchange rate is...
-
What are the potential off-target effects associated with CRISPR-Cas9 genome editing, and what strategies are being developed to enhance the specificity and reduce unintended consequences ?
-
Justice seeks to make things right. The idea of justice is based on ethics, equality, human rights, religion, and solidarity. In an exploration of the work of John Rawls, Otried, ed. (2014: Section...
-
What does it mean when it is said, as a political scientist, one should 'never get angry at a fact'? 2. Define comparative politics. Discuss what comparative politics entails and why it is important...
-
Explain how base editing and prime editing, as modifications of the CRISPR-Cas9 system, differ in their mechanisms and potential applications compared to traditional double-strand break-mediated...
-
I think I am missing some steps because I can not come up with this answer. can you please explain? Takeshi KamadaCIA Japan (A). Takeshi Kamada, a foreign exchange trader at Credit Suisse (Tokyo), is...
-
Suppose that the laptop of Prob. 2.16 is placed in an insulating briefcase with a fully charged battery, but it does not go into sleep mode, and the battery discharges as if the laptop were in use....
-
Isopropanol is prepared by reacting propylene (CH3CHCH2) with sulfuric acid, followed by treatment with water. (a) Show the sequence of steps leading to the product. What is the role of sulfuric...
-
Using the standard reduction potentials listed in Table 19.1 and the Handbook of Chemistry and Physics, show that the following reaction is favorable under standard-state conditions: What is the...
-
One of the steps in the extraction of iron from its ore (FeO) is the reduction of iron(II) oxide by carbon monoxide at 900C: FeO(s) + CO(g) Fe(s) + CO2(g) If CO is allowed to react with an excess of...
-
Develop the formula (10.43) for the case of the \(x\)-momentum equation of a two-dimensional steady-state incompressible flow discretized on a structured uniform finite difference grid. Use central...
-
Show that the decomposition (10.37) of the nonlinear term is correct. Use direct substitution of (10.36) into the expression for one component of vector \(N\).
-
For the flow in Problem 7, write the boundary conditions for pressure when the flow is incompressible and inviscid and there is no body force.

Study smarter with the SolutionInn App


