Oxygen is composed of three isotopes: oxygen-16, oxygen-17 and oxygen-18 and has an average atomic mass...
Fantastic news! We've Found the answer you've been seeking!
Question:
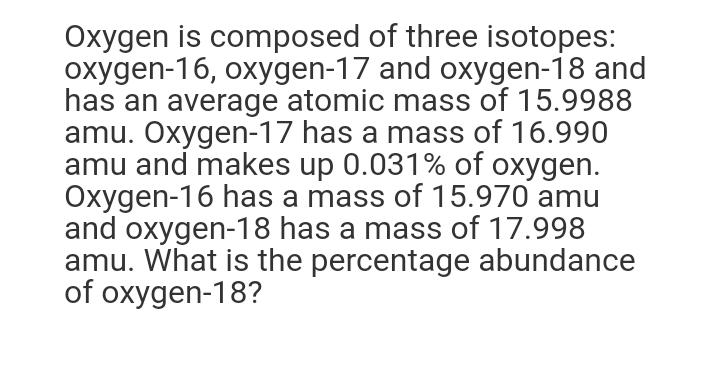
Transcribed Image Text:
Oxygen is composed of three isotopes: oxygen-16, oxygen-17 and oxygen-18 and has an average atomic mass of 15.9988 amu. Oxygen-17 has a mass of 16.990 amu and makes up 0.031% of oxygen. Oxygen-16 has a mass of 15.970 amu and oxygen-18 has a mass of 17.998 amu. What is the percentage abundance of oxygen-18? Oxygen is composed of three isotopes: oxygen-16, oxygen-17 and oxygen-18 and has an average atomic mass of 15.9988 amu. Oxygen-17 has a mass of 16.990 amu and makes up 0.031% of oxygen. Oxygen-16 has a mass of 15.970 amu and oxygen-18 has a mass of 17.998 amu. What is the percentage abundance of oxygen-18?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
15) Pb has an average atomic mass of 207.19amu. The three major isotopes of Pb are Pb-206(205.98amu); Pb-207(206.98 amu) and Pb-208(207.98 amu). If the isotopes of Pb-207 and Pb-208 are present in...
-
Chlorine has an average atomic mass of 35.45 amu. The two naturally occurring isotopes of chlorine are chlorine-35 and chlorine-37. Why does this indicate that most chlorine atoms contain 18 neutrons?
-
The transition temperature of a BCS superconductor with an average atomic mass of 199.5 is 4.185k. what is the transition temperature for an average atomic mass of 203.4?
-
1. Learn the detail background on ERP systems. Discuss how you would integrate ERP systems internal and external across your entire organization or an organization of your choice? Then discuss the...
-
Watson Corporation's comparative balance sheet for current assets and liabilities was as follows: Adjust net income of $320,000 for changes in operating assets and liabilities to arrive at net cash...
-
Which pair will form a compound with the larger lattice energy: Na and F or Mg and F? Why?
-
PhotoMax, Inc., plans to prepare a sales budget schedule and a purchases budget schedule for 20X3. Management has set a sales goal of 250,000 units. After reviewing price trends, the sales manager...
-
The loan department of Local Bank uses standard costs to determine the overhead cost of processing loan applications. During the current month a fire occurred, and the accounting records for the...
-
Project L requires an initial outlay at t 0 of $80,097, its expected cash inflows are $14,000 per year for 9 years, and its WACC is 13%. What is the project's IRR? Round your answer to two decimal...
-
Rianni Sonns recently died after a brief illness and is survived by her two children, Daysha and Dominic. Rianni was very close with her daughter, Daysha, but had not had a relationship with her son...
-
1. Write down the mathematical model and Draw the block diagram then Obtain the transfer function Vo(s)/V/(s) of the electrical systems shown in Figure (1). Where V(s) denotes the input voltage and...
-
You want to purchase a bond that has 5.6667years(five years and eight months)left to maturity.It pays interest semiannually and has a coupon rate of 4.8% and a yield to maturity of 3.9%.Its face...
-
Factoring a small number, such as 36, may be pretty simple, but how might one go about factoring a larger number, such as 19,800?
-
a. Equipment with a book value of $65,300 and an original cost of $133,000 was sold at a loss of $14,000. b. Paid $89,000 cash for a new truck. c. Sold land costing $154,000 for $198,000 cash,...
-
Lamda corporation wants to acquire another company within its industry for $100m and it expects the acquisition to contribute to its free cash flow by $5m the first year, and this contribution is...
-
Discuss the concept of value proposition, positioning strategy, customer satisfaction and retention and their relationship to organizational and marketing success. Explain Briefly?
-
Discuss a time when language barriers or a lack of cross-cultural training became a challenge at your organization. What challenge did it present at work, and how could the organization have improved...
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
a. Is the reaction of 2-butene with HBr regioselective? b. Is it stereoselective? c. Is it stereospecific? d. Is the reaction of 1-butene with HBr regioselective? e. Is it stereoselective? f. Is it...
-
Give the products of the following reactions. (Hint: See Problem 67.) a. b. Br HO CH3
-
The bond angles in H3O+ are greater than __________ and less than __________.
-
A service enterprise performs services in the amount of \(\$ 500\) for a customer in May and receives payment in June. In which month is the \(\$ 500\) of revenue recognized? What is the journal...
-
Late in March, Classic Auto Painters purchased paint on account, with payment due in 60 days. The company used the paint to paint customers' cars during the first three weeks of April. Late in May,...
-
How does the accrual basis of accounting differ from the cash basis of accounting? Which gives a more accurate picture of the profitability of a business? Explain.

Study smarter with the SolutionInn App



