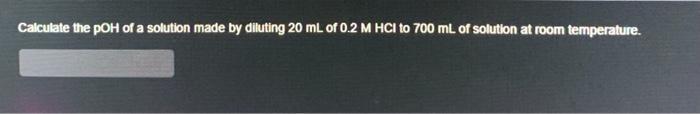
Calculate the pOH of a solution made by diluting 20 mL of 0.2 M HCI to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the pOH of a solution made by diluting 20 mL of 0.2 M HCI to 700 mL of solution at room temperature. Calculate the pOH of a solution made by diluting 20 mL of 0.2 M HCI to 700 mL of solution at room temperature.
Expert Answer:
Answer rating: 100% (QA)
Initial Concentration of HCl M1 02M V1 20mL After dilut... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the standard enthalpy change for the reaction of an aqueous solution of barium hydroxide, Ba(OH)2, with an aqueous solution of ammonium nitrate, NH4NO3, at 25oC. (Figure 6.1 illustrated...
-
Calculate the standard enthalpy change for the reaction 2Al(s) + Fe2O3(s) - 2Fe(s) + Al2O3(s) given that 2Al(s) +3O2(g)- Al2O3(s) n_-1669 - F603(s) .8 mol 2Fe(s) + O2(g) -MR00-822.2 kJ/mol 2(g)
-
At 298 K, the standard enthalpies of formation for C2H2(g) and C6H6(l) are 227 kJ/ mol and 49 kJ/ mol, respectively. a. Calculate Ho for C6H6(l) 3C2H2(g) b. Both acetylene (C2H2) and benzene (C6H6)...
-
Car A is due north of Car B. In the Earth frame, Car A is moving south and Car B is moving East. What direction is Car B moving as seen by Car A? north south east west north and east north and west...
-
A glassblower makes glass decanters and glass trays on a weekly basis. Each item requires 1 pound of glass, and the glassblower has 15 pounds of glass available each week. A glass decanter requires 4...
-
Explain what ethical issues SOX has helped with and where improvement is still needed.
-
1. Identify 3 students to play the roles of the employees. Ask these 3 individuals to read their roles below. 2. Identify 1 student to play the role of the president of the social enterprise (Taylor...
-
Chapman Company, a major retailer of bicycles and accessories, operates several stores and is a publicly traded company. The comparative balance sheet and income statement for Chapman as of May 31,...
-
35. Expandiendo e sin en serie de Fourier en wt, mostrar que la ecuacin trascendedente de Kepler tiene la solucin formal: = wt+Jn(ne) sin (wt), n=1 n 2 donde J, es la funcin de Bessel de orden n....
-
Griffin and Lasky, Inc. (G&L), supplies industrial automation equipment and machine tools to the automotive industry. G&L recognizes revenue on its long-term contracts over time. Customer orders have...
-
Read the scenario: $80 in 1hr. 1page REAL LIFE...It's Monday and Professor Malone has just assigned a 10-page research paper on Personal Skills. You have one week to complete the paper and it counts...
-
Simplify the following expression by expanding and collecting like terms. 10(-3)+6(-8x+3) Give your answer in the form az + b.
-
Journalize Period Payroll The payroll register of Chen Engineering Co. indicates $2,220 of social security withheld and $555 of Medicare tax withheld on total salaries of $37,000 for the period....
-
Rhett Corporation manufactures and sells dress shirts. Each shirt (unit) requires 2 yards of cloth. Selected data from Rhett's master budget for next quarter are shown below: April May June Budgeted...
-
Find log5,log4, and log20. Show that log20=log5+log4. What property of logarithms does this equality illustrate? What property of exponentiation does this property come from?
-
The actual cost of direct labor per hour is $8.00, and the budgeted cost of direct labor per hoour is $7.50. Three budgeted direct labor hours are allowed for each finished unit. During the current...
-
Your PE firm has decided to invest $0.8 million in a startup, in return for a 30% share of the firm's equity. What's the implied (post-money) valuation of the firm, based on that offer? Answer in...
-
Q:1 Take any product or service offered in Pakistan and apply all determinents of customer Perceived value ?
-
In a murder trial in the 1990s, the defendant's blood was found at the crime scene. The prosecutor argued that blood was left by the defendant during the crime. The defense argued that police...
-
(a) Neglecting activity coefficients and ion pairing, find the pH and concentrations of species in 1.00 L of solution containing 0.040 mol 2-aminobenzoic acid (a neutral molecule, HA), 0.020 mol...
-
The observed behavior of benzyl alcohol (C6H5CH2OH) in capillary electrophoresis is given here. Draw a graph showing the number of plates versus the electric field and explain what happens as the...
-
The purchasing officer for The Majestic Emporium has prepared a purchases budget for the financial year ending 31 March 2020, based on the following data. The cost of sales is 65% of sales, and the...
-
Greyt Dog Beds Ltd manufactures dog beds specifically designed for greyhounds and whippets. The company purchases all the required materials from external suppliers and designs and assembles the...
-
Ketton Ltd is in the process of preparing direct labour and factory overhead budgets for the year ending 30 June 2020. Relevant data are set out below. Required (a) Prepare the direct labour budget...

Study smarter with the SolutionInn App


