Based off of the conceptual reading, when comparing eyes open state to eyes closed, when would...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
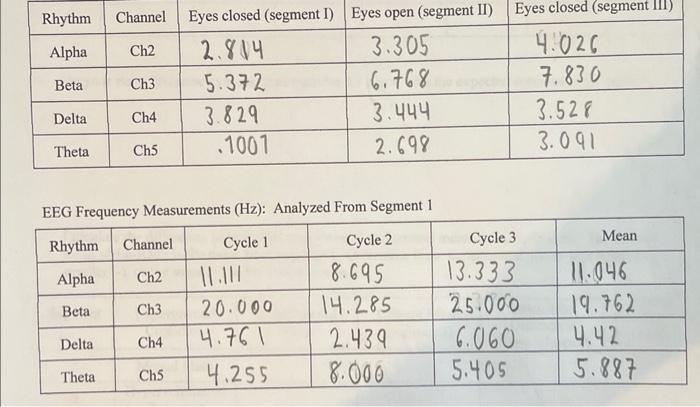
Based off of the conceptual reading, when comparing eyes open state to eyes closed, when would the beta rhythm be most pronounced? (Explain) Did your data reflect this? Based off of the conceptual reading, is there an increase or decrease in delta and theta wave activity when the eyes are open? (Explain) Did your data reflect this? Rhythm Channel Eyes closed (segment I) Eyes open (segment II) Eyes closed (segment III) Alpha 2.814 3.305 4.026 Beta 5.372 6.768 7.830 3.829 3.444 2.698 Delta Theta Ch2 Delta Ch3 Theta Ch4 Ch5 EEG Frequency Measurements (Hz): Analyzed From Segment 1 Cycle 1 Cycle 2 Rhythm Channel Alpha Ch2 Beta Ch3 Ch4 .1001 Ch5 20.000 4.761 4.255 8.695 14.285 2.439 8.000 Cycle 3 13.333 25.000 6.060 5.405 3.528 3.091 Mean 11.046 19.762 4.42 5.887 Based off of the conceptual reading, when comparing eyes open state to eyes closed, when would the beta rhythm be most pronounced? (Explain) Did your data reflect this? Based off of the conceptual reading, is there an increase or decrease in delta and theta wave activity when the eyes are open? (Explain) Did your data reflect this? Rhythm Channel Eyes closed (segment I) Eyes open (segment II) Eyes closed (segment III) Alpha 2.814 3.305 4.026 Beta 5.372 6.768 7.830 3.829 3.444 2.698 Delta Theta Ch2 Delta Ch3 Theta Ch4 Ch5 EEG Frequency Measurements (Hz): Analyzed From Segment 1 Cycle 1 Cycle 2 Rhythm Channel Alpha Ch2 Beta Ch3 Ch4 .1001 Ch5 20.000 4.761 4.255 8.695 14.285 2.439 8.000 Cycle 3 13.333 25.000 6.060 5.405 3.528 3.091 Mean 11.046 19.762 4.42 5.887
Expert Answer:

Posted Date:
Students also viewed these chemical engineering questions
-
Q. Calculate the amount of dividends the company declared during fiscal 2021. Your answer should be your numeric calculation, in equation form. What number of shares did the company use in the...
-
The bicarbonate ion has the ability to act as an acid in the presence of a base and as a base in the presence of an acid, so it is said to be amphiprotic. Illustrate this behavior with water by...
-
An unknown mass of Al absorbs 187.9 J of heat and increases its temperature from 23.5(C to 35.6(C. What is the mass of the aluminum? How many moles of aluminum is this?
-
Sketch a graph to illustrate your idea of each of the following relationships. Be sure to label each axis appropriately. For each relationship, explain under what circumstances, if any, the curve...
-
Beginning inventory, purchases, and sales data for hammocks are as follows: a. Assume the business maintains a perpetual inventory system, costing by the first-in, first-out method. Determine the...
-
Chris Lane, the head professional at Royal Oak Country Club, must develop a schedule of matches for the couples golf league that begins its season at 4:00 P.M. tomorrow. Eighteen couples signed up...
-
Two very long wires each carry a linear charge density \(\lambda\). They initially repel each other with a force \(F\). If the wires are immersed in distilled water, with what force do they repel...
-
Reporting Cash Basis versus Accrual Basis Income Payson Sports, Inc., sells sports equipment to customers. Its fiscal year ends on December 31. The following transactions occurred in 2012: a....
-
1. What is Stock Market Analysis. Explain in Detail? 2. Discuss about Nature and Scope of Stock Market Analysis. 3. Discuss about Basics of Stock Market Analysis. 4. What is Investment? Explain in...
-
1. Prepare the journal entries for transactions. (If no entry is required for a particular transaction/event, select "No journal entry required" in the first account field.) 2. Prepare an income...
-
A person wants to know whether the average waiting time for restaurant A is longer than the average waiting time of restaurant B. Assume the population variances are equal. What statistical method...
-
What is an asset-backed security?
-
What is the distinguishing feature of how municipal bonds are taxed?
-
Six particles in a container can move in only one of six directions: up, down, left, right, forward, and backward. If the particles move randomly such that each collides and changes direction every...
-
To a vessel that contains \(1.00 \mathrm{~mol}\) of monatomic ideal gas \(\mathrm{A}\) is added \(0.100 \mathrm{~mol}\) of monatomic ideal gas \(\mathrm{B}\). The mass of each atom of gas A is \(3.35...
-
What is an efficient market?
-
Electric charge q= -4 mC and mass m= 3.1 kg moves with a speed v= 8 km/s along the electric field (with it or against it). What is the speed of the charge after the charge travels through a potential...
-
Doorharmony Company makes doorbells. It has a weighted- average cost of capital of 5% and total assets of $ 5,900,000. Doorharmony has current liabilities of $ 750,000. Its operating income for the...
-
Refer to the latest financial report of JB Hi-Fi Limited on its website, www.jbhifi.com.au, and answer the following questions. 1. Is it likely that JB Hi-Fi Limited would have to confront such...
-
Imelda Instruments Ltd manufactures two products: missile range instruments and space pressure gauges. During January, 53 range instruments and 360 pressure gauges were produced, and overhead costs...
-
Swiss Chocolates Ltd produces blocks of chocolate. Raw materials in the form of cocoa solids, milk and sugar are added at the beginning of the process, flavouring, fruit and nuts are added half-way...

Study smarter with the SolutionInn App

