Part 1. Density of water 1. Volume of water. 2. Mass of beaker: 3. Mass of...
Fantastic news! We've Found the answer you've been seeking!
Question:




Transcribed Image Text:
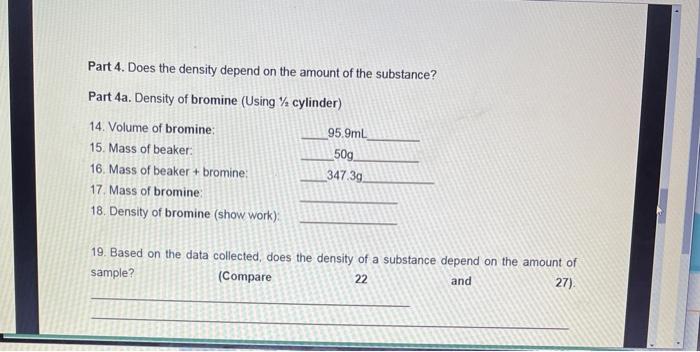
Part 1. Density of water 1. Volume of water. 2. Mass of beaker: 3. Mass of beaker + water: 4. Mass of water: 5. Density of water (show work): Part 2. Density of gasoline 6. Volume of gasoline: 7. Mass of beaker: 8. Mass of beaker + gasoline: 9. Mass of gasoline: 10. Density of gasoline (show work). 50mL 50g 100g 85mL 50g 116.3g Part 3. Miscibility of liquids Miscible liquids completely combine to form one solution. Immiscible liquids do not mix completely (heterogeneous mixture) Fill a graduated cylinder with " water and ½ gasoline" 11. Are 12. Which water and gasoline miscible or immiscible? liquid is at the top? Which liquid is at the bottom? 13. Is this expected from your previously collected data? Explain in terms of density. (Compare 5 and 10) Part 4. Does the density depend on the amount of the substance? Part 4a. Density of bromine (Using ½ cylinder) 14. Volume of bromine: 15. Mass of beaker: 16. Mass of beaker + bromine: 17. Mass of bromine: 18. Density of bromine (show work): 95.9mL 50g 347.3g 19. Based on the data collected, does the density of a substance depend on the amount of sample? (Compare 22 and 27). Part 6. Density of copper by displacement of liquid (water) 20. Mass of copper: 21. Volume of water: 22. Volume of water + metal: 23. Volume of copper: 24. Density of copper (show work): Part 7. Additional problems 50g 50ml 55.8mL 25. A ball of material "X" has a mass of 10.830 g and occupies a volume of 19 mL. What is the density of material "X"? 26. What will happen to a ball of material "X" if placed in a container with gasoline? Will the ball of material "X" float or sink? Explain in terms of density. Part 1. Density of water 1. Volume of water. 2. Mass of beaker: 3. Mass of beaker + water: 4. Mass of water: 5. Density of water (show work): Part 2. Density of gasoline 6. Volume of gasoline: 7. Mass of beaker: 8. Mass of beaker + gasoline: 9. Mass of gasoline: 10. Density of gasoline (show work). 50mL 50g 100g 85mL 50g 116.3g Part 3. Miscibility of liquids Miscible liquids completely combine to form one solution. Immiscible liquids do not mix completely (heterogeneous mixture) Fill a graduated cylinder with " water and ½ gasoline" 11. Are 12. Which water and gasoline miscible or immiscible? liquid is at the top? Which liquid is at the bottom? 13. Is this expected from your previously collected data? Explain in terms of density. (Compare 5 and 10) Part 4. Does the density depend on the amount of the substance? Part 4a. Density of bromine (Using ½ cylinder) 14. Volume of bromine: 15. Mass of beaker: 16. Mass of beaker + bromine: 17. Mass of bromine: 18. Density of bromine (show work): 95.9mL 50g 347.3g 19. Based on the data collected, does the density of a substance depend on the amount of sample? (Compare 22 and 27). Part 6. Density of copper by displacement of liquid (water) 20. Mass of copper: 21. Volume of water: 22. Volume of water + metal: 23. Volume of copper: 24. Density of copper (show work): Part 7. Additional problems 50g 50ml 55.8mL 25. A ball of material "X" has a mass of 10.830 g and occupies a volume of 19 mL. What is the density of material "X"? 26. What will happen to a ball of material "X" if placed in a container with gasoline? Will the ball of material "X" float or sink? Explain in terms of density.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A piston cylinder contains 1 kg of liquid water at 20oC and 300 kPa. There is a linear spring mounted on the piston such that when the water is heated the pressure reaches 3 MPa with a volume of 0.1...
-
Show that a mixture of saturated liquid water and saturated water vapor at 300 kPa satisfies the criterion for phase equilibrium.
-
A cylinder with radius R and mass M has density that increases linearly with distance r from the cylinder axis, p = ar, where a is a positive constant (a) Calculate the moment of inertia of the...
-
At fiscal year-end December 31, 2015, Shop-World had the following assets and liabilities on its balance sheet (in millions): Current liabilities ............ $9,459 Long-term debt .................
-
You learn that the Dow Jones Total Stock Market market-value-weighted index increased by 16 percent during a specified period, whereas a Dow Jones Total Stock Market equal-weighted index increased by...
-
Describe why the observational approach and predictive design is the most appropriate methodology to be used considering the research scenario. Develop a hypothetical research scenario that would...
-
How are computers and digital devices used in cybercrime?
-
Sanderson Manufacturing produces ornate, decorative wood frame doors and windows. Each item produced goes through 3 manufacturing processes: cutting, sanding, and finishing. Each door produced...
-
Suppose the consumer has a wage of $52.00 per week to spend on Goods A, B & C. The cost per unit of each of these goods is Good A: $1.00 per unit, Good B: $2.00 per unit, Good C: $4.00 per unit. Use...
-
On December 31, the Income Summary account of Johnson Company has a Credit balance of $226,000 after revenue of $519,000 and expenses of $293,000 were closed to the account. Gloria Johnson, Drawing...
-
In thermodynamics entropy is the heat transferred into a system divided by the temperature, or if Q is the heat transferred, then we say S = Q/T. The "Delta" means change in. Entropy was a focus of...
-
Zap Industries had a tentative general business credit in 2018 of \(\$ 60,000\). Its tax liability in 2017 was \(\$ 20,000\). Its tax liability before the general business credit in 2018 is \(\$...
-
An external confirmation sent to a customer: a. provides strong evidence for the accuracy, valuation, and allocation assertion when the customer agrees with the balance owing. b. requests information...
-
During the following years Adolpho T. (single) had (1) withheld, (2) paid (by check - presumably estimated tax payments or paid with the extension) and (3) received a refund of the following amounts...
-
Why do most companies underforecast than overforecast? Cite a situation when overforecasting may be better.
-
The assertion of cut-off: a. reflects that all assets and liabilities that should have been recorded are recorded. b. ensures that events have been recorded in the correct accounts. c. is an...
-
Young has gone on to accumulate other credit card debt on top of what he owes from his Disney Land vacation and his total debit is now $13,864.82. He is getting worried about his debt and is...
-
Read the Forecasting Supply Chain Demand Starbucks Corporation case in your text Operations and Supply Chain Management on pages 484-485, then address the four questions associated with the...
-
A mixture having a volume of 10.00 mL and containing 0.1000 M Ag + and 0.100 0 M Hg 2 2+ was titrated with 0.1000 M KCN to precipitate Hg 2 (CN) 2 and AgCN. (a) Calculate pCN - at each of the...
-
Consider the titration of 100.0 mL of 0.010 0 M Ce 4+ in 1 M HClO 4 by 0.040 0 M Cu + to give Ce 3+ and Cu 2+ , using Pt and saturated Ag | AgCl electrodes to find the end point. (a) Write a balanced...
-
I have always enjoyed eating tuna fish. Unfortunately, a study of the mercury content of canned tuna in 2010 found that chunk white tuna contains 0.6 ppm Hg and chunk light tuna contains 0.14 ppm....
-
Show that, in Theorem 19.17, the quadratic variation \(\langle Mangle_{t}\) is a \(\mathscr{G}_{t}\) stopping time.Direct calculation, use Lemma 19.16.c) and A. 15 Data From Theorem 19.17 19.17...
-
The proof of Theorem 19.29 uses, implicitly, the following beautiful result due to Skorokhod [239] which is to be proved: Lemma. Let \(b:[0, \infty) ightarrow \mathbb{R}\) be a continuous function...
-
We have seen in Lemma 19.27.a) that \(\operatorname{supp}\left[d L_{t}^{0}(\omega) ight] \subset\left\{t \geqslant 0: B_{t}(\omega)=0 ight\}\) for almost all \(\omega\). Show that...

Study smarter with the SolutionInn App


