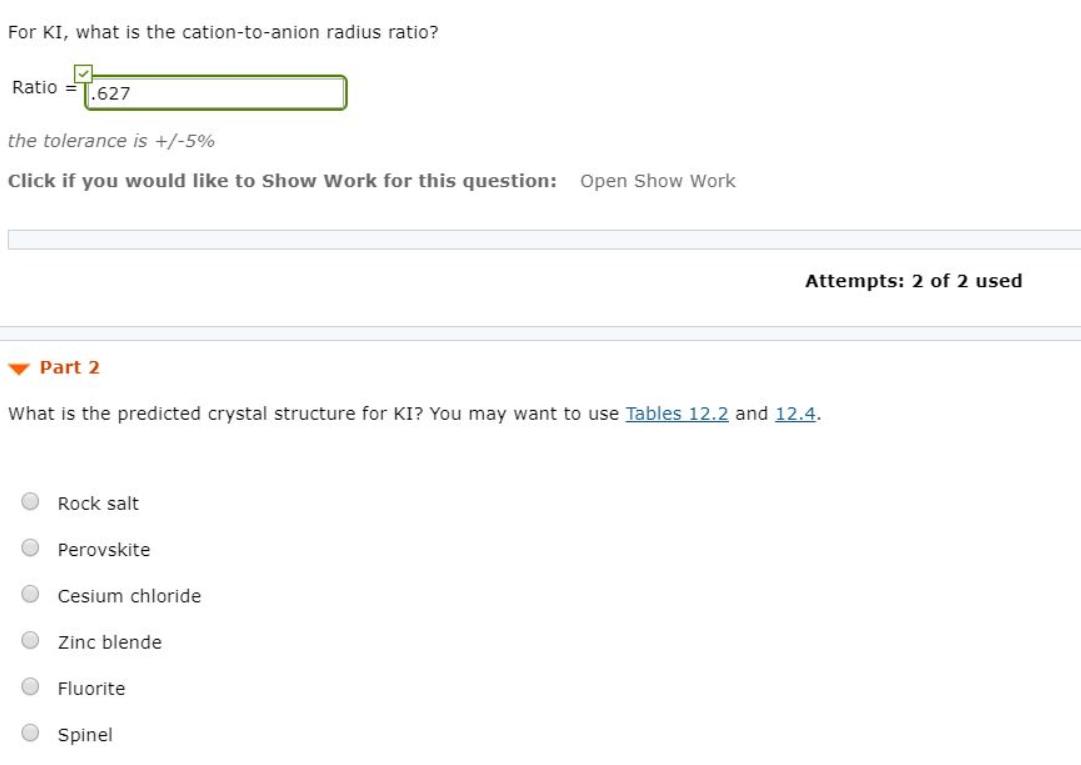
For KI, what is the cation-to-anion radius ratio? Ratio .627 the tolerance is +/-5% Click if...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For KI, what is the cation-to-anion radius ratio? Ratio .627 the tolerance is +/-5% Click if you would like to Show Work for this question: Open Show Work Part 2 What is the predicted crystal structure for KI? You may want to use Tables 12.2 and 12.4. Rock salt Perovskite Cesium chloride Zinc blende Fluorite Attempts: 2 of 2 used Spinel For KI, what is the cation-to-anion radius ratio? Ratio .627 the tolerance is +/-5% Click if you would like to Show Work for this question: Open Show Work Part 2 What is the predicted crystal structure for KI? You may want to use Tables 12.2 and 12.4. Rock salt Perovskite Cesium chloride Zinc blende Fluorite Attempts: 2 of 2 used Spinel
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Part 1 Question What is the cationtoani... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
what is the cation ?? 9. Cation whose sulphide salt is canary yellow ppt. 10. Cation escapes from group IV to group III 11. Cation from group Il soluble in dil HNO3 and forms a white precipitate with...
-
The zinc blende crystal structure is one that may be generated from close-packed planes of anions. (a) Will the stacking sequence for this structure be FCC or HCP? Why? (b) Will cations fill...
-
Would you use an anion or cation exchange column to purify bovine histone at pH 7.0?
-
Revonce Company sells merchandise on a consignment basis to dealers .The selling price of the merchandise averages 25% above cost. The dealer is paid a 10% commission of the sales price for all sales...
-
Downhill Boards (DB), a producer of snow boards, is evaluating a new process for applying the finish to its snow boards. Durable Finish Company (DFC) has offered to apply the finish for $170,000 in...
-
The balance sheet items of Mercer Company as of December 31, 2011, follow in random order. You are to prepare a balance sheet for the company, using a similar sequence for assets as illustrated in...
-
What is the primary purpose of admission-seeking questions?
-
You have been assigned to the first audit of the Chicago Company for the year ending March 31, 2011. Accounts receivable were confirmed on December 31, 2010, and at that date the receivables...
-
quick question are they same " the function of database management system and the characteristics of database management system? explain maintaining data integrity and give simple explanation to...
-
4. Distinct Items There is a list of items in the shopping cart, each having a cost associated with it. There are n items, the cost of the ith item is / dollars and m items have already been bought...
-
You are the manager of an educational facility and have beenasked to predict analyze the electric cost for 2020 based on the following 12 months of Information, You know the electric cost has both...
-
Given the following diagram: a) Prove that AABC is similar to AADE ABC is ZB of the large triangle. Middle letter indicates the angle within the triangle indicated using the three letters. 6 cm b)...
-
Exercise 4. Company XYZ is evaluating the feasibility of manufacturing a new product. The plant it owns has the capacity to produce 1,000,000 units of the product. Fixed costs for this initiative...
-
Skippy wants to have $17,000.00 in 10 years. His bank is offering an account that earns 1% compounded monthly. How much does he need to deposit to reach his goal? Round your final answer up to the...
-
Rachel wants to have $3,600.00 in 36 months. Her bank is offering her a Certificate of Deposit, a special savings account, that earns 2.3% compounded weekly. How much does she need to deposit now to...
-
Fresh Ltd has two retail businesses that represent separate cash generating units, Fresh Juice Bar and Fresh Salads. At 30 June 2019, the carrying amounts of the assets of the units, valued pursuant...
-
Tom needs to purchase a new car urgently. Unfortunately he has no funds at the moment, but his grandmother has promised to give him $75,000 on his birthday in six months. How much can Frank...
-
Which of the following gives the range of y = 4 - 2 -x ? (A) (- , ) (B) (- , 4) (C) [- 4, ) (D) (- , 4] (E) All reals
-
You have a factory with 40 production machines that are essentially identical, each producing at a mean daily rate of 90 products with a standard deviation of 35. You may assume that they produce...
-
How are prices set for computer processor chips? At one time, the frequency was a good indicator of the processing speed; however, more recently manufacturers have developed alternative ways to...
-
High salaries for presidents and high executives of charitable organizations have been in the news from time to time. Consider the information in Table 11.3.13 for the United Way in 10 major cities....
-
A blender does 5000 J of work on the food in its bowl. During the time the blender runs, 2000 J of heat is transferred from the warm food to the cooler environment. What is the change in the thermal...
-
Which system contains more atoms: 5 mol of helium (A = 4) or 1 mol of neon (A = 20)? A. Helium B. Neon C. They have the same number of atoms.
-
A sample of ideal gas is in a sealed container. The temperature of the gas and the volume of the container are both increased. What other properties of the gas necessarily change? (More than one...

Study smarter with the SolutionInn App


