Part A Describe the hybridization of the carbon atom in the poisonous gas phosgene, Cl CO....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
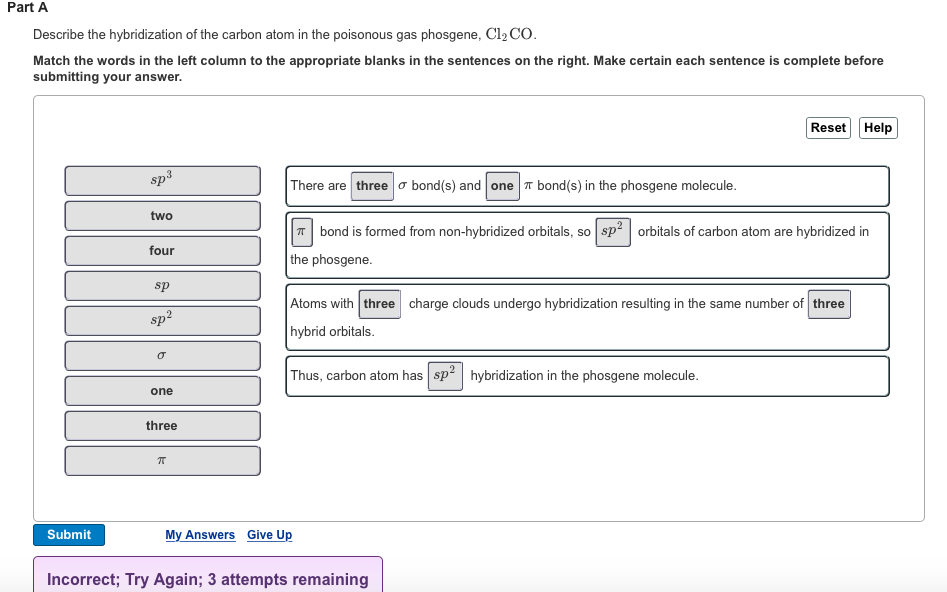
Part A Describe the hybridization of the carbon atom in the poisonous gas phosgene, Cl₂ CO. Match the words in the left column to the appropriate blanks in the sentences on the right. Make certain each sentence is complete before submitting your answer. Submit sp³ two four sp 2 sp² σ one three π There are three bond(s) and one bond(s) in the phosgene molecule. bond is formed from non-hybridized orbitals, so sp² orbitals of carbon atom are hybridized the phosgene. Reset Help Atoms with three charge clouds undergo hybridization resulting in the same number of three hybrid orbitals. Thus, carbon atom has sp² hybridization in the phosgene molecule. My Answers Give Up Incorrect; Try Again; 3 attempts remaining Part A Describe the hybridization of the carbon atom in the poisonous gas phosgene, Cl₂ CO. Match the words in the left column to the appropriate blanks in the sentences on the right. Make certain each sentence is complete before submitting your answer. Submit sp³ two four sp 2 sp² σ one three π There are three bond(s) and one bond(s) in the phosgene molecule. bond is formed from non-hybridized orbitals, so sp² orbitals of carbon atom are hybridized the phosgene. Reset Help Atoms with three charge clouds undergo hybridization resulting in the same number of three hybrid orbitals. Thus, carbon atom has sp² hybridization in the phosgene molecule. My Answers Give Up Incorrect; Try Again; 3 attempts remaining
Expert Answer:
Answer rating: 100% (QA)
C1 0 FCI Therefore there are three sigma bonds 0 and one pi bond 7 in the ... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
Describe the hybridization of each carbon atom in each of the following structures. Base your answer on the geometry about the carbon atom. (a) CH3CI (b) CH3OH (c) CH3CH2CH3 (d) CH2 == CH2 (trigonal...
-
Predict the hybridization of the carbon atom in each of the following functional groups: (a) Ketone (b) Nitrile (c) Carboxylic acid
-
What is the hydribization of each carbon atom in acetonitrile (Problem 1.25)?
-
The 2015 income statement of Adrian Express reports sales of $19,310,000, cost of goods sold of $12,250,000, and net income of $1,700,000. Balance sheet information is provided in the following...
-
Fechter Corporation had the following stockholders equity accounts on January 1, 2014: Common Stock ($5 par) $500,000, Paid-in Capital in Excess of Par Common Stock $200,000, and Retained Earnings...
-
In 1940, the family of Thomas Back entered into an oil-and-gas lease with the Inland Gas Corporation. The lease held that Inland would pay to Backs family 12 cents per thousand cubic feet of gas...
-
The inventory of Wood4Fun and data on purchases and sales for a two-month period follow. The company closes its books at the end of each month. It uses the periodic inventory system. Required 1....
-
Prove that for all x = R, if a, b = Z such that x = a + b2, then a and b are unique.
-
Julio and Milania are owners of Falcons Corporation, an S corporation. They each own 50 percent of Falcons Corporation. In year 1, Julio and Milani received distributions of $20,000 and $10,000,...
-
(4.00 Puanlar) 2- Which of the following lines are used to fill the blank parts of the following code block for the purpose of combining two singly link list by appending list2 to list1. void...
-
LINUX operating system does not suffer serious deadlocks like windows operating systems. Explain the disadvantages of:Linux operating systems and windows operating systems?
-
Compare two differences and two similarities between a desktop operating system versus a network operating system. Include file structure and boot process in your comparison. Limit your discussion to...
-
King Fisher Aviation purchases from suppliers in a quarter are equal to 65% of the next quarter's forecast sales. The payables period is 30 days. Wages, taxes, and other expenses are 25% of sales,...
-
Suppose that you sell for $14 a call option with a strike price of $47, sell for $7 a call option with a strike price of $57, and buy for $9 each two call options with a strike price of $52. What is...
-
Last year Carson Industries issued a 10-year, 12% semiannual coupon bond at its par value of $1,000. Currently, the bond can be called in 6 years at a price of $1,060 and it sells for $1,150. What is...
-
You are trying to value a private company. The company has 4 million of debt and 4 million of book equity. The ratio of market value to book value for similar firms is 2. You decide to use this ratio...
-
What is the order p of a B + -tree? Describe the structure of both internal and leaf nodes of a B + -tree.
-
For each of the following groups, rank the molecules in decreasing order of reactivity toward addition of a nucleophile to the most electrophilic sp2 -hybridized carbon. (a) (b) (c) BrCH 2 COCH 3 ,...
-
Formulate the mechanism of the BF3-catalyzed reaction of CH 3 SH with butanal.
-
Illustrate the results of chain elongation of D-talose through a cyanohydrin. How many products are formed? Draw them. After treatment with warm HNO 3 , does the product(s) give optically active or...
-
Management is considering three alternatives to satisfy an urgent need. Each of the alternatives will completely satisfy the need, so no combinations have to be considered. The first costs, operating...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/ year. a. Determine the MIRR for this project. b. Is this project economically...

Study smarter with the SolutionInn App


