Part A Give the mass of the solute and the volume of the solvent for 1.70...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
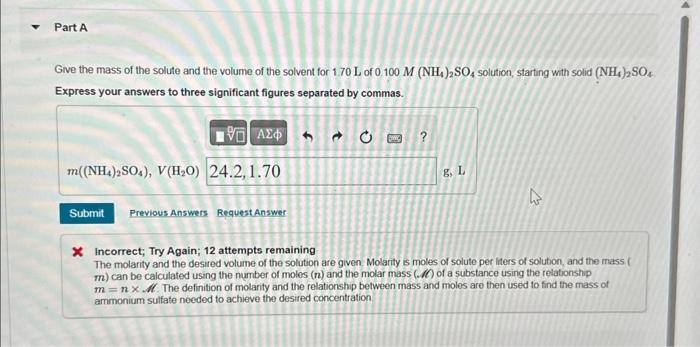
Part A Give the mass of the solute and the volume of the solvent for 1.70 L of 0.100 M (NH4)2SO4 solution, starting with solid (NH4)2SO4 Express your answers to three significant figures separated by commas. 195 ΑΣΦρά ο m((NH,),SO4), V(H,O) 24.2,1.70 Submit Previous Answers Request Answer ? 8, L 4 * Incorrect; Try Again; 12 attempts remaining The molarity and the desired volume of the solution are given. Molarity is moles of solute per liters of solution, and the mass ( m) can be calculated using the number of moles (n) and the molar mass (.) of a substance using the relationship m = nxM. The definition of molarity and the relationship between mass and moles are then used to find the mass of ammonium sulfate needed to achieve the desired concentration ► Part A Give the mass of the solute and the volume of the solvent for 1.70 L of 0.100 M (NH4)2SO4 solution, starting with solid (NH4)2SO4 Express your answers to three significant figures separated by commas. 195 ΑΣΦρά ο m((NH,),SO4), V(H,O) 24.2,1.70 Submit Previous Answers Request Answer ? 8, L 4 * Incorrect; Try Again; 12 attempts remaining The molarity and the desired volume of the solution are given. Molarity is moles of solute per liters of solution, and the mass ( m) can be calculated using the number of moles (n) and the molar mass (.) of a substance using the relationship m = nxM. The definition of molarity and the relationship between mass and moles are then used to find the mass of ammonium sulfate needed to achieve the desired concentration ► Part A Give the mass of the solute and the volume of the solvent for 1.70 L of 0.100 M (NH4)2SO4 solution, starting with solid (NH4)2SO4 Express your answers to three significant figures separated by commas. 195 ΑΣΦρά ο m((NH,),SO4), V(H,O) 24.2,1.70 Submit Previous Answers Request Answer ? 8, L 4 * Incorrect; Try Again; 12 attempts remaining The molarity and the desired volume of the solution are given. Molarity is moles of solute per liters of solution, and the mass ( m) can be calculated using the number of moles (n) and the molar mass (.) of a substance using the relationship m = nxM. The definition of molarity and the relationship between mass and moles are then used to find the mass of ammonium sulfate needed to achieve the desired concentration ►
Expert Answer:
Answer rating: 100% (QA)
To solve this problem we need to calculate both the mass of the solute NH42SO4 ammonium sulfate and ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Constraints show the product's total cycle time or limit throughput: True? The target cost is determined by taking the difference between market price and desired profit. True? The sales life cycle...
-
Having just returned from serving with the U.S. Marines in Afghanistan, Nick has managed to save most of his earnings. He has enrolled in the local community college and he is back living with his...
-
Under a plan of complete liquidation, Coast Corporation distributes land with a $300,000 adjusted basis and a $400,000 FMV to William, a 25% shareholder. William has a $200,000 basis in his Coast...
-
The Sourdough Bread Company also allocates fixed manufacturing overhead to products on the basis of standard direct manufacturing labor-hours. For 2017, fixed manufacturing overhead was budgeted at...
-
In the circuit of Problem 9.59 find phasor currents I 1 and I 2 drawn from the two sources. Data from Problem 9.59 In the circuit of Fig. 9-55, v 1 = 6 cos t and v 2 = cos (t + 60). Find v A if = 2...
-
Sweet Bayou Airlines (SBA) flight 494 departs daily from Shreveport for Nashville at 8:47 a.m. The manager of the SBA office in Shreveport must receive a file that combines to contents of the file...
-
At the end of 2008, Activo Company implemented a low-cost strategy to improve its competitive position. Its objective was to become the low-cost producer in its industry. A Balanced Scorecard was...
-
Discuss how the Eastern market will potentially view new hire orientation, and then discuss two to three ways to tailor the new hire orientation to this market?
-
It is July 2022, and all the figures relating to your budgets and actual trading performance have been assembled: Sales were 10% above projection. Cost of Goods Sold came in 6% over expectation. ...
-
Illustrate using a single diagram (graph). Illustrate using a single diagram (ie.graph) Assume that the U.S. inflation rate becomes high relative to Canadian inflation. Other things being equal, how...
-
Watch the Tedx Talk (Melvin Russell: I love being a police officer, but we need reform) by Lt. Colonel Melvin Russel from the Baltimore Police. In his talk, he discusses how law enforcement is in...
-
How do different thread scheduling algorithms (e.g., round-robin, priority-based, and fair-share) influence the responsiveness and resource utilization of a multithreaded system ?
-
Dr. Daniela Yeung, a health psychologist, has been conducting a federally funded ethnographic study of couples in which the male partner has been imprisoned for intimate partner violence. Over the...
-
Suppose you have the following processes Process PI P2 P3 P4 Arrival Time 0 Burst Time 14 2 10 3 2 5 7 7 13 P5 Use the following multilevel feedback queue CPU scheduling algorithm to execute the...
-
Explain what is meant by the term gross negligence (IN YOUR OWN WORDS) and provide four examples of situations in sports in which the conduct of a defendant might rise to this level of negligence....
-
On September 1, 2020, Oriole Company reacquired 30400 shares of its $10 par value common stock for $15 per share. Oriole uses the cost method to account for treasury stock. The journal entry to...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
When methanol,CH3OH, is dissolved in water, a nonconducting solution results. When acetic acid,CH3COOH, dissolves in water, the solution is weakly conducting and acidic in nature. Describe what...
-
A quantity of the blue solid on the left in Figure 13.7 is placed in an oven and heated for a time. It slowly turns from blue to the white of the solid on the right. What has occurred?
-
Halons are fluorocarbons that contain bromine, such as CBrF3. They are used extensively as foaming agents for fighting fires. Like CFCs, halons are very unreactive and ultimately can diffuse into the...
-
When randomly selecting a day of the week, it is certain that you will select a day containing the letter y, so P(y) = 1. For Exercises 510, decide whether the statement makes sense (or is clearly...
-
Use subjective judgment to estimate the probability that the next time you ride an elevator, it gets stuck between floors.
-
Because it is impossible for Thanksgiving to fall on Tuesday, the probability of Thanksgiving falling on Tuesday is 0. For Exercises 510, decide whether the statement makes sense (or is clearly true)...

Study smarter with the SolutionInn App


