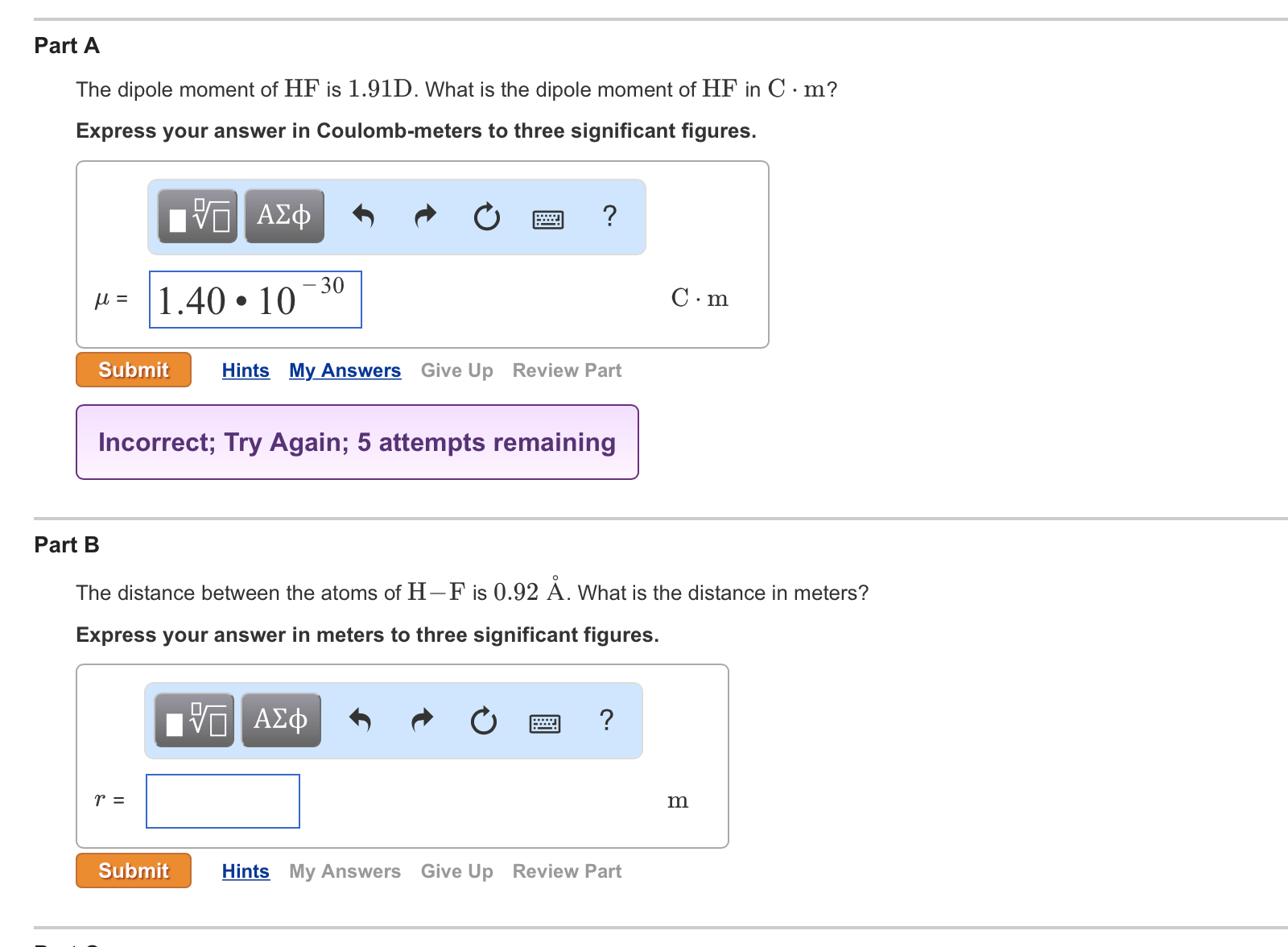
Part A The dipole moment of HF is 1.91D. What is the dipole moment of HF...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Part A The dipole moment of HF is 1.91D. What is the dipole moment of HF in C. m? Express your answer in Coulomb-meters to three significant figures. Τ— ΑΣΦ με 1.40 10 -30 Submit Hints My Answers Give Up Review Part r = wwwww Incorrect; Try Again; 5 attempts remaining ? ΓΠ ΑΣΦ Submit Part B The distance between the atoms of H-F is 0.92 Å. What is the distance in meters? Express your answer in meters to three significant figures. ? C.m Hints My Answers Give Up Review Part m Part A The dipole moment of HF is 1.91D. What is the dipole moment of HF in C. m? Express your answer in Coulomb-meters to three significant figures. Τ— ΑΣΦ με 1.40 10 -30 Submit Hints My Answers Give Up Review Part r = wwwww Incorrect; Try Again; 5 attempts remaining ? ΓΠ ΑΣΦ Submit Part B The distance between the atoms of H-F is 0.92 Å. What is the distance in meters? Express your answer in meters to three significant figures. ? C.m Hints My Answers Give Up Review Part m
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The dipole moment of the water molecule (H20) is 6.17 X 10-30 C. m. Consider a water molecule located at the origin whose dipole moment p points in the + x-direction. A chlorine ion (C1-), of charge...
-
Given the dipole moment of water ( I .84 D) and the H-O-H bond angle (104.45o), justify the statement in Problem 1.43(b) that the bond dipole moment of the O-H bond is 1.52 D.
-
(a) Show that the dipole moment of l,4-dioxane (Sec. 8.1C) should be zero if the molecule exists solely in a chair conformation, (b) Account for the fact that the dipole moment of 1,4- dioxane,...
-
Bryce owns 200 shares of Basic Company stock that he purchased for $8,000 three years ago. On December 28, 2021, Bryce sold 100 shares of the stock for $2,500. On January 3, 2022, Bryce repurchased...
-
Shaw Company revealed the following information for the years 2009 and 2010: Shaw Company Income Statement For the Year Ended December 31, 2010 Sales ..........$ 55,000 Cost of goods sold...
-
A stock is selling for $100 with a volatility of 40 percent. Consider a call option on the stock with an exercise price of 100 and an expiration of one year. The risk-free rate is 4.5 percent. Let...
-
Which of the following Assertion is inaccurate concerning the auditors inquiry regarding fraud? (a) Inquiries of management concerning its knowledge of fraud, alleged fraud, or suspected fraud...
-
Anna Broderick is the dietitian for the State University football team, and she is attempting to determine a nutritious lunch menu for the team. She has set the following nutritional guidelines for...
-
Kailash an auditor, is the engagement partner on the financial statement audit of Super Computer Services Bhd (SCS) for the year ended 30 April 2019. On 5 May 2019, the senior auditor assigned to...
-
The State of Delaware is the corporate haven of the United States. More than 50 percent of the publicly traded corporations in America, including 60 percent of the Fortune 500 companies, are...
-
Sunland Company uses a perpetual inventory system and reports the following for the month of June. Date Explanation Units Unit Cost Total Cost June 1 Inventory 140 $5 $700 12 Purchase 360 2,160 23...
-
12) Find the inverse f(x) of each. a) f(x)=9+10x x b) f(x)=- 7x+2
-
2-2 Find a basis of the column space of the matrix -31 A = -4 -7 7 10 -2 E M3x5 (R): 5 7 -6 -10 1
-
You are required to analyse a listed company and make a investment recommendation report. The report provides an assessment of the company's current position and future prospects, incorporating the...
-
Question 1: Please transform the following E-R diagram to relations (20 points). (Note: Dep_Names means the dependent names for each Client.) E_Type DOB E_Phone E_ID Age Schedule EMPLOYEE "S"...
-
A 5 0 , 0 0 0 - pound truck is parked on a 1 0 you slope ( see figure ) . Assume the only force to overcome is that due to gravity. ( Round your answers to one decimal place. ) Weight = 5 0 , 0 0 0...
-
1) Our investigation of Tutoring hours per week (x) and math SAT score (y) shows a Linear relationship between the two Variables. For Tutoring hours, the average (
-
(a) How far away can a human eye distinguish two ear headlights 2.0 m apart? Consider only diffraction effects and assume an eye pupil diameter of 5.0 mm and a wavelength of 550 nm. (b) What is the...
-
Arrange the following oxides in order of increasing acidity: CO 2 , CaO,Al 2 O 3 , SO 3 , SiO 2 , and P 2 O 5 .
-
Sulfur dioxide reacts with strontium oxide as follows: SO2(g) + SrO(s) SrSO3(s) (a) Without using thermochemical data, predict whether G for this reaction is more negative or less negative than H....
-
The degradation of CF3CH2F (an HFC) by OH radicals in the troposphere is first order in each reactant and has a rate constant of k = 1.6 108 M-1 s-1 at 4oC. If the tropospheric concentrations of OH...
-
A friend of yours tells you that his parents plan to pay for his college education by selling some family jewelry. Write About It Write a paragraph explaining why you might question this plan.
-
Explain why so many fraud complaints are related to online auctions.
-
Imagine that you have \($10,000\) to invest. Use a newspaper, the Internet, or other source to select a mutual fund. Use the same source to fi nd out how much gold you could buy for \($10,000\)....

Study smarter with the SolutionInn App



