Part A Watch the animation and identify the correct conditions for forming a hydrogen bond. Check...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
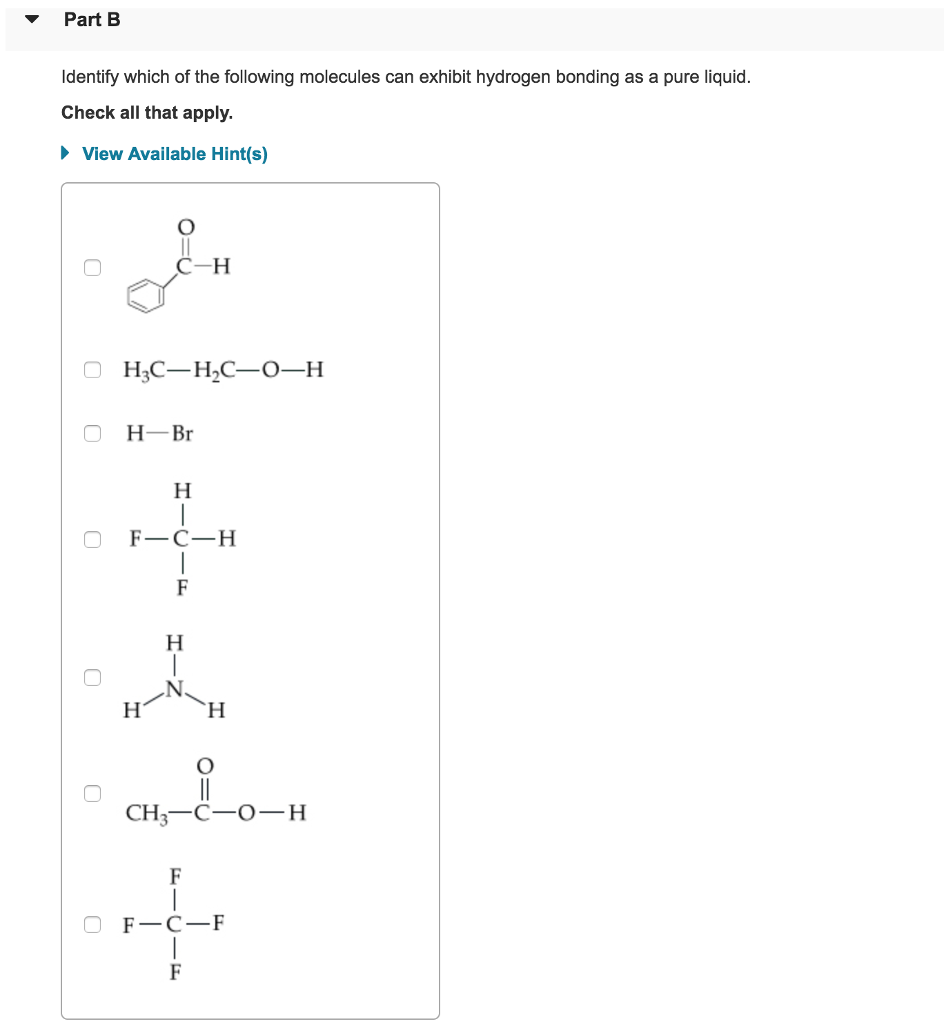
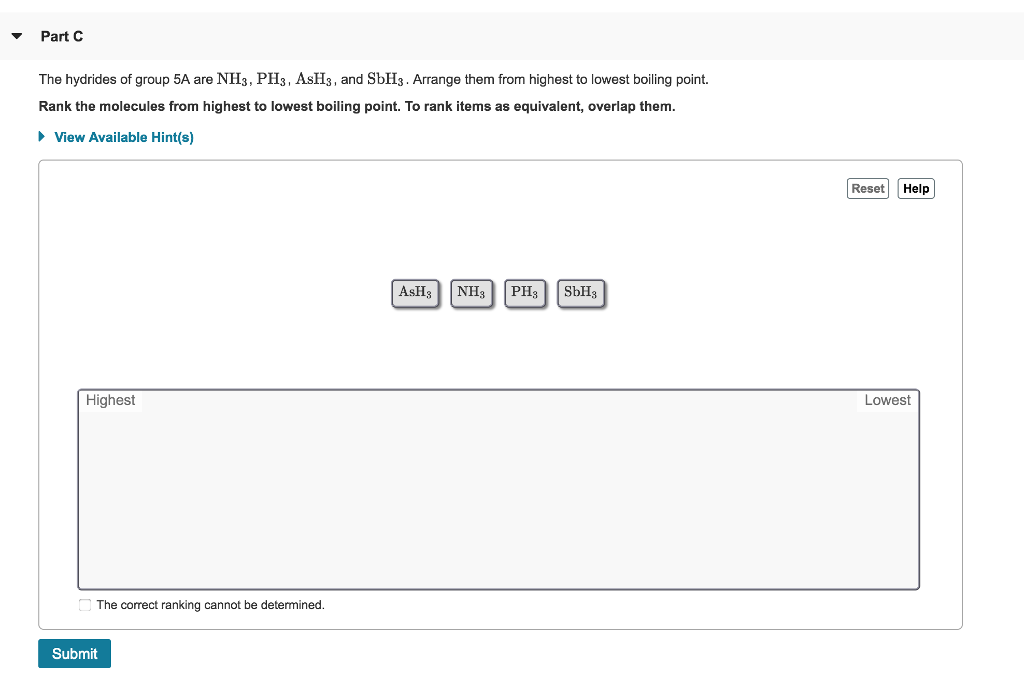
Part A Watch the animation and identify the correct conditions for forming a hydrogen bond. Check all that apply. ► View Available Hint(s) ооооо The CH4 molecule exhibits hydrogen bonding. A hydrogen bond is possible with only certain hydrogen-containing compounds. Hydrogen bonding occurs when a hydrogen atom is covalently bonded to an N, O, or F atom. A hydrogen atom acquires a partial positive charge when it is covalently bonded to an F atom. A hydrogen bond is equivalent to a covalent bond. Submit Part B Identify which of the following molecules can exhibit hydrogen bonding as a pure liquid. Check all that apply. ► View Available Hint(s) 0 OH₂C-H₂C-0-H 0 0 O H-Br H H F-C-H F H H F O CH3-C-0-H H F OF-C-F Part C The hydrides of group 5A are NH3, PH3, ASH3, and SbH3. Arrange them from highest to lowest boiling point. Rank the molecules from highest to lowest boiling point. To rank items as equivalent, overlap them. ▸ View Available Hint(s) Highest The correct ranking cannot be determined. Submit AsH₂ NH3 PH3 SbH3 Reset Help Lowest Part A Watch the animation and identify the correct conditions for forming a hydrogen bond. Check all that apply. ► View Available Hint(s) ооооо The CH4 molecule exhibits hydrogen bonding. A hydrogen bond is possible with only certain hydrogen-containing compounds. Hydrogen bonding occurs when a hydrogen atom is covalently bonded to an N, O, or F atom. A hydrogen atom acquires a partial positive charge when it is covalently bonded to an F atom. A hydrogen bond is equivalent to a covalent bond. Submit Part B Identify which of the following molecules can exhibit hydrogen bonding as a pure liquid. Check all that apply. ► View Available Hint(s) 0 OH₂C-H₂C-0-H 0 0 O H-Br H H F-C-H F H H F O CH3-C-0-H H F OF-C-F Part C The hydrides of group 5A are NH3, PH3, ASH3, and SbH3. Arrange them from highest to lowest boiling point. Rank the molecules from highest to lowest boiling point. To rank items as equivalent, overlap them. ▸ View Available Hint(s) Highest The correct ranking cannot be determined. Submit AsH₂ NH3 PH3 SbH3 Reset Help Lowest
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following molecules may be polar? (a) CH3Cl (C3J, (b) HW2 (CO)10 (D4h), (c) SnCl4 (Td).
-
Which of the following molecules may show a pure rotational microwave absorption spectrum? (a) H20, (b) H202, (c) NH4 (d) N20?
-
Which of the following molecules may show a pure rotational Raman spectrum? (a) CH2Cl2 (b) CH3CH3, (c) SP6, (d) N2O?
-
The people on Coral Island buy only juice and cloth. The CPI market basket contains the quantities bought in 2016. The average household spent $60 on juice and $30 on cloth in 2016 when the price of...
-
Figure includes a box for the econometrics unit. Item (3) is to help other units. What sorts of specific tasks might thisentail? Customers Product Final Risky Portfolio Feedback Overall Portfolio...
-
Suppose compartments A and B shown in the following figure are filled with fluidsand are separated by a permeable membrane. The figure is a compartmental representation of the exterior and...
-
Identify at least five sources for locating the whereabouts of a witness.
-
Zeidman Security Services Co offers security services to business clients. The trial balance for Zeidman Security Services Co. has been prepared on the end-of-period spreadsheet (work sheet) for the...
-
Capitalized Lease - Part of FINAL HW assignment Lena Leasing Ltd. Leases excavating equipment that has a list price of $300,000 to Morgan Mining Co. on January 1, 2022. Ownership of the equipment...
-
Refer to the financial statements of The Home Depot in Appendix A at the end of this book. Required: 1. How much did The Home Depots sales revenue increase or decrease in the year ended January 29,...
-
Regulation of individual professional accountants is primarily conducted at a national level, with professional accountancy organizations playing an important role in working with governments to...
-
Describe the five characteristics of an effective VOCAL delivery (vocal elements) and illustrate each one with an example that will be the most challenging for you to master. What will you do...
-
What are the seven types of verbal supporting materials? For each, provide at least one principle of effective use. Which is your favorite supporting material? Why?
-
Jones Manufacturing Inc. sponsored a defined benefit pension plan effective 1 January 20X7. The company uses the projected unit credit actuarial cost method for funding and accounting. Long-term...
-
(a) Explain the difference between an Euler path and an Euler cycle. (b) Find the maximum number of comparisons to be made to find any record in a binary search tree which holds 3000 records. [3] (c)...
-
Explain the terms 'risk disclosure' and' 'no guarantee of performance, in the context of financial advice.
-
b) A deflection type spring balance for measuring mass is calibrated in an environment at a temperature of 20C. When used at 40C there will be some error in the measured values according to the...
-
On the basis of the details of the following fixed asset account, indicate the items to be reported on the statement of cashflows: ACCOUNT Land ACCOUNT NO. Balance Date Item Debit Credit Debit Credit...
-
Aspartame is an artificial sweetener that is 160 times sweeter than sucrose (table sugar) when dissolved in water. It is marketed as NutraSweet. The molecular formula of aspartame is C 14 H 18 N 2 O...
-
The element europium exists in nature as two isotopes: 151 Eu has a mass of 150.9196 amu, and 153 Eu has a mass of 152.9209 amu. The average atomic mass of europium is 151.96 amu. Calculate the...
-
A certain sample of uranium is reacted with fluorine to form a mixture of 235 UF6(g) and 238 UF 6 (g). After 100 diffusion steps, the gas contains 1526 235 UF 6 molecules per 1.000 10 5 total number...
-
Identify a company that integrates goods and services similar to the LensCrafters example. Use the framework in Exhibit 5.1 to write a short case study about the company in a similar fashion as the...
-
Airbags are designed to protect passengers from frontal or near-frontal crashes. Their designs and performance have improved dramatically since their introduction in the 1980s. Exhibit 5.19 shows a...
-
Glen County manages a waste-to-energy facility that burns 2,000 tons of trash per day and generates over \($20\) million in electricity annually while costing state and local taxpayers \($24\)...

Study smarter with the SolutionInn App


