A solution of water (Kf point of pure water is 0.00 C. = 1.86 C/m) and...
Fantastic news! We've Found the answer you've been seeking!
Question:
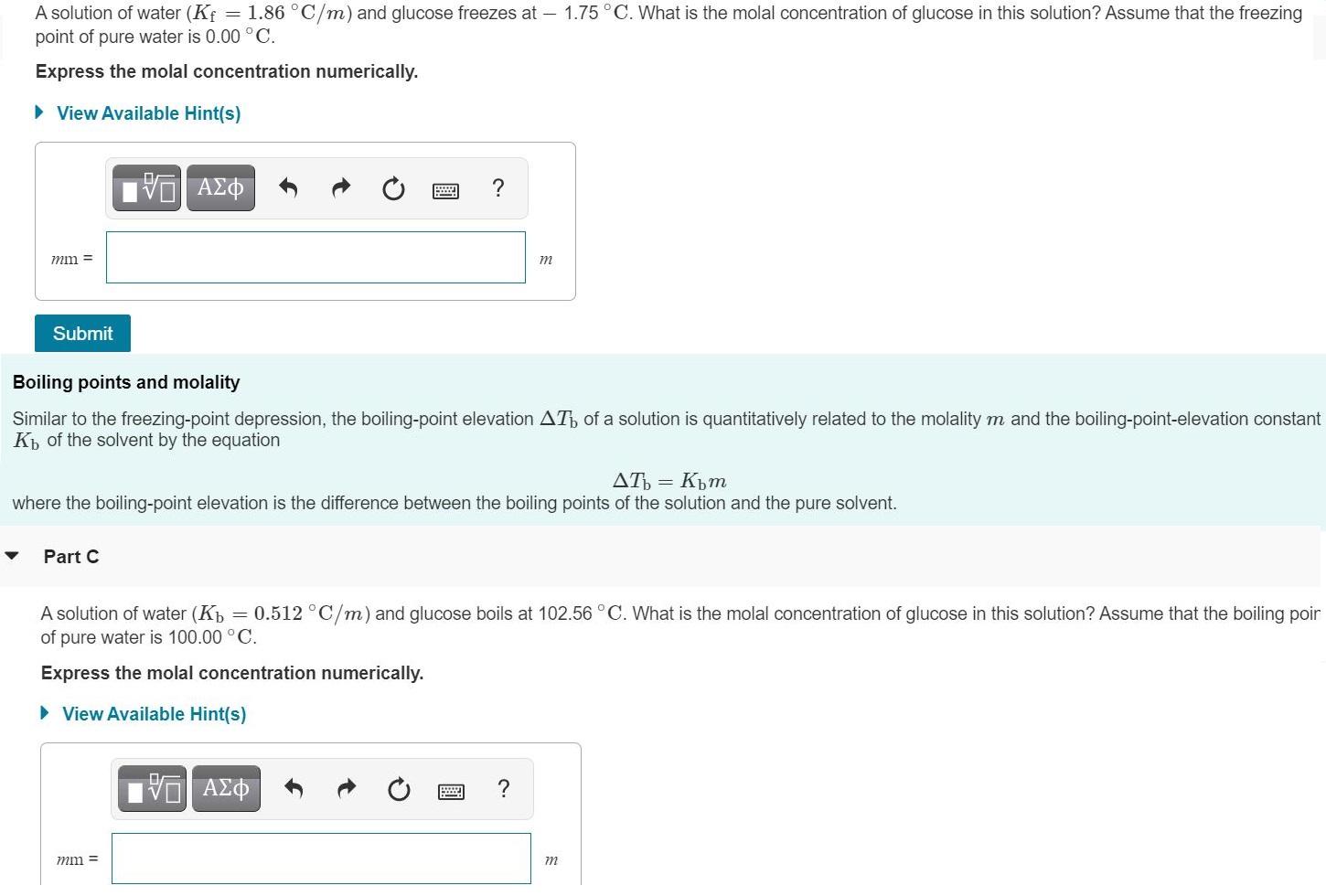
Transcribed Image Text:
A solution of water (Kf point of pure water is 0.00 °C. = 1.86 °C/m) and glucose freezes at – 1.75°C. What is the molal concentration of glucose in this solution? Assume that the freezing Express the molal concentration numerically. • View Available Hint(s) mm = m Submit Boiling points and molality Similar to the freezing-point depression, the boiling-point elevation AT of a solution is quantitatively related to the molality m and the boiling-point-elevation constant K of the solvent by the equation AT = Kpm where the boiling-point elevation is the difference between the boiling points of the solution and the pure solvent. Part C A solution of water (Kp = 0.512 °C/m) and glucose boils at 102.56°C. What is the molal concentration of glucose in this solution? Assume that the boiling poir of pure water is 100.00 °C. Express the molal concentration numerically. • View Available Hint(s) mm = m A solution of water (Kf point of pure water is 0.00 °C. = 1.86 °C/m) and glucose freezes at – 1.75°C. What is the molal concentration of glucose in this solution? Assume that the freezing Express the molal concentration numerically. • View Available Hint(s) mm = m Submit Boiling points and molality Similar to the freezing-point depression, the boiling-point elevation AT of a solution is quantitatively related to the molality m and the boiling-point-elevation constant K of the solvent by the equation AT = Kpm where the boiling-point elevation is the difference between the boiling points of the solution and the pure solvent. Part C A solution of water (Kp = 0.512 °C/m) and glucose boils at 102.56°C. What is the molal concentration of glucose in this solution? Assume that the boiling poir of pure water is 100.00 °C. Express the molal concentration numerically. • View Available Hint(s) mm = m
Expert Answer:

Related Book For 

Practical Management Science
ISBN: 978-1305250901
5th edition
Authors: Wayne L. Winston, Christian Albright
Posted Date:
Students also viewed these chemistry questions
-
Refer to Decision Maker, Purchase Manager, in this chapter. Assume that you are the motorcycle manufacturers managerial accountant. The purchasing manager asks you about preparing an estimate of the...
-
Refer to Decision Maker, Purchase Manager, in this chapter. Assume that you are the motorcycle manufacturers managerial accountant. The purchasing manager asks you about preparing an estimate of the...
-
For the problems in this exercise, assume that there are no pipeline stalls and that the breakdown of executed instructions is as follows: 1. In what fraction of all cycles is the data memory used?...
-
Preparing and interpreting a statement of cash flows using a T-account work sheet. Financial statement data for Dickerson Manufacturing Company for the current year appear in Exhibit 5.29. Additional...
-
The gas law for an ideal gas at absolute temperature T (in kelvins), pressure P (in atmospheres), and volume V (in liters) is PV = nRT, where is the number of moles of the gas and R = 0.0821 is the...
-
Rank from fastest to slowest the following waves according to their speed of propagation: A. An earthquake wave B. A tsunami C. A sound wave in air D. A light wave
-
Why is an ICP accelerometer better than a non-ICP accelerometer?
-
Stonewall Company was organized on January 1, 2015. During the year ended December 31, 2015, the company provided the following data: Analysis of selected accounts and transactions follows: a. Issued...
-
A metallic sphere of diameter 5 cm is initially at a uniform temperature of 300oC. Suddenly the metal sphere is immersed in a fluid at 30oC. The convective heat transfer coefficient between the...
-
You are responsible for the audit of inventory for Honey Best Grocery Wholesales, Inc., a closely held grocery wholesaler that sells to independent grocery stores. Inventory is by far the largest...
-
After three centuries under the rule of Portugal, Brazil became an independent nation in 1822. By far the largest and most populous country in South America, with a population of more than 205...
-
Imagine that an acquaintance of yours was recently arrested for shoplifting a $15 necktie. Write an explanation for this behavior from each perspective on abnormality: the medical perspective, the...
-
__________is defined as a response to challenging or threatening events.
-
Distinguish between financial accounting and managerial accounting.
-
Use the following information to answer the below questions pertaining to the adjusting entries of Afaaq Company and Noor Takaful. Afaaq Company purchased an insurance policy for $5,000 on December...
-
Match the disorder with the culture in which it is most common: a. Amok b. Anorexia nervosa c. Brain fag Catatonic schizophrenia India b. Malaysia a. United States d. West Africa C.
-
The expansion of Martha Harper's franchising idea today drives half of all retail sales in america. This means one woman has had a remarkable effect on america's economy. So why is she relatively...
-
Illini Company, Inc. Balance Sheet as of 12/31/20X0 Assets Current Assets: Cash $1,500,000 Accounts receivable, net 18,000 Inventory 50,000 Total current assets 1,568,000 Equipment 90,000 Goodwill...
-
A beer company has divided Bloomington into two territories. If the company spends x 1 dollars on promotion in territory 1, it can sell 60x 1 1/2 cases of beer there; and if it spends x 2 dollars on...
-
Use Solver Table to run a sensitivity analysis on the cost per assembling labor hour, letting it vary from $5 to $20 in increments of $1. Keep track of the computers produced in row 21, the hours...
-
Do the sales prices of houses in a given community vary systematically with their sizes (as measured in square feet)? Answer this question by estimating a simple regression equation where the sales...
-
James Halperin, co-chair of Heritage Auctions, was interviewed about his experiences auctioning rare coins. Noting that when the auction house first opened in the 1970s, I used to proofread every...
-
An editorial on Bloomberg. com criticized President Bidens Buy American policy. The editorial argued that the . . . problem is that protectionism sets the US at odds with its international...
-
An article in the Wall Street Journal explained that despite rising national security tensions between the United States and China in 2023, trade between the two nations was rising. China is a...

Study smarter with the SolutionInn App


