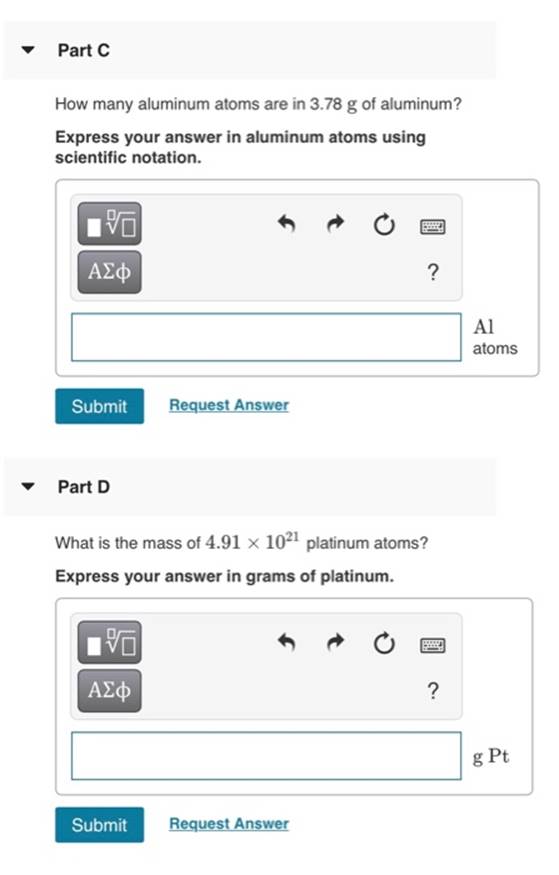
Part C How many aluminum atoms are in 3.78 g of aluminum? Express your answer in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Part C How many aluminum atoms are in 3.78 g of aluminum? Express your answer in aluminum atoms using scientific notation. ΑΣφ ? Al atoms Submit Request Answer Part D What is the mass of 4.91 x 1021 platinum atoms? Express your answer in grams of platinum. ΑΣφ ? g Pt Submit Request Answer Part C How many aluminum atoms are in 3.78 g of aluminum? Express your answer in aluminum atoms using scientific notation. ΑΣφ ? Al atoms Submit Request Answer Part D What is the mass of 4.91 x 1021 platinum atoms? Express your answer in grams of platinum. ΑΣφ ? g Pt Submit Request Answer
Expert Answer:
Answer rating: 100% (QA)
Answer Past C As wrdas mass of Al 26981 u Griven mase of Af 3789 Molee f Al 348g O 14 moles 26... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
What is the mass of a 1.00-Ci 14/8C source?
-
What is the mass of the solid NH4Cl formed when 73.0 g of NH3 are mixed with an equal mass of HCl? What is the volume of the gas remaining, measured at 14.0C and 752 mmHg? What gas is it?
-
What is the mass density of air at P = 1.0 atm and T = (a) 10 C and (b) 30 C? The average molecular mass of air is approximately 29 u.
-
Common structural steel can be represented on drawings using a letter, immediately followed by a number, then another number (for example, S14x30). The two numbers for a steel member W16x60 designate...
-
Now that you have read the chapter on health care planning, what do you recommend to Danielle DiMartino in the case at the beginning of the chapter regarding: 1. Choosing among the four alternatives...
-
Dr. Meredith Grey is a 76-year-old woman that presented to the outpatient clinic for reports of problems with attention/concentration (e.g., problems focusing for long periods and is easily...
-
What is the role of the Auditing Practices Board in the UK regulatory framework? Discuss whether it has been successful in achieving its objectives.
-
Dropping a customer, activity-based costing, ethics. Jack Arnoldson is the management accountant for Valley Restaurant Supply (VRS). Bob Gardner, the VRS sales manager, and Jack are meeting to...
-
How do external factors, such as economic conditions or industry trends, influence organizational commitment, and how should organizations adapt to these influences ?
-
John Wallace is an automotive enthusiast. He has over 25 years of experience as a mechanic for the dealership of a large car manufacturer in Oakville. John also gained experience doing minor body...
-
You believe there is a 60% chance of recession and 40% chance of expansion next year. If the annual return on PJ Nickel is -3% in a recession and 5% in an expansion, what do you expect the return on...
-
Continue with the case of Shiva Industries Ltd. as above. In the beginning of the 6th year, a valuer appraises the machine to be worth Rs. 97.50 lakh with an estimated residual value of Rs. 4,87,500....
-
This case is in continuation to the case of Oil and Natural Gas Corporation Ltd. as illustrated in the last chapter. Now refer further to the extracts from annual report 2005-06 of the company...
-
It can be interesting to compare the purchasing power of the dollar over different periods in history. Go to www.bankofcanada.ca/en/rates.htmand scroll down to the link to the inflation calculator to...
-
For the lid-driven cavity problem, conduct parametric studies for \(R e=1, R e=100, R e=200\). Discuss, qualitatively, how the flow field changes with the \(R e\).
-
Go to the Web site of the Office of National Statistics (https:// www.ons.gov.uk/), the United Kingdom's producer of official data and check the section "Employment and labor market." a. How does the...
-
Question 4: In analogy to transesterification, transamidation is a reaction where one amide is converted into another in the presence of an amine nucleophile. Transamidation is typically a very slow...
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
Making mayonnaise involves beating oil into small droplets in water, in the presence of egg yolk. What is the purpose of the egg yolk?
-
Acetic acid is a polar molecule and can form hydrogen bonds with water molecules. Therefore, it has a high solubility in water. Yet acetic acid is also soluble in benzene (C6H6), a nonpolar solvent...
-
Acetic acid is a weak acid that ionizes in solution as follows: If the freezing point of a 0.106 m CH 3 COOH solution is 20.203C, calculate the percent of the acid that has undergone ionization....
-
In an audit of a corporation that has a bond issue outstanding, the trust indenture is reviewed and confirmation as to the issue is obtained from the trustee. List eight matters of importance to the...
-
Robertson Company had accounts receivable of \(\$ 200,000\) at December 31, 200X, and had provided an allowance for uncollectible accounts of \(\$ 6,000\). After performing all normal auditing...
-
Tom Jones, CPA, is auditing the financial statements of a manufacturing company with a significant amount of trade accounts receivable. Jones is satisfied that the accounts are properly summarized...

Study smarter with the SolutionInn App


