Question: Piechocki Corporation manufactures and sells a single product. The company uses units as the measure of activity in its budgets and performance reports. During
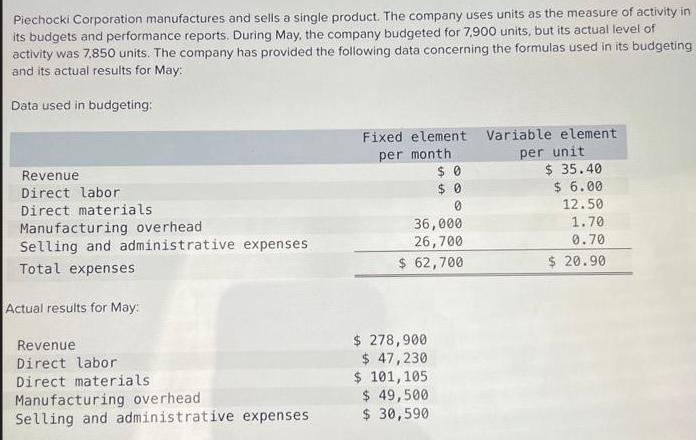

Piechocki Corporation manufactures and sells a single product. The company uses units as the measure of activity in its budgets and performance reports. During May, the company budgeted for 7,900 units, but its actual level of activity was 7,850 units. The company has provided the following data concerning the formulas used in its budgeting and its actual results for May: Data used in budgeting: Revenue Direct labor Direct materials Manufacturing overhead Selling and administrative expenses Total expenses Actual results for May: Revenue Direct labor Direct materials Manufacturing overhead Selling and administrative expenses. Fixed element per month $0 $0 0 36,000 26,700 $ 62,700 $ 278,900 $ 47,230 $ 101, 105 $ 49,500 $ 30,590 Variable element. per unit $ 35.40 $ 6.00 12.50 1.70 0.70 $20.90 Calculate the revenue variance for May
Step by Step Solution
3.41 Rating (151 Votes )
There are 3 Steps involved in it
Calculate the revenue variance for May Given data Budgeted units 7900 units Actual units 78... View full answer

Get step-by-step solutions from verified subject matter experts


