Please answer this two question. D Try Again Your answer is incorrect. C3 S: Your answer
Fantastic news! We've Found the answer you've been seeking!
Question:
Please answer this two question.

Transcribed Image Text:
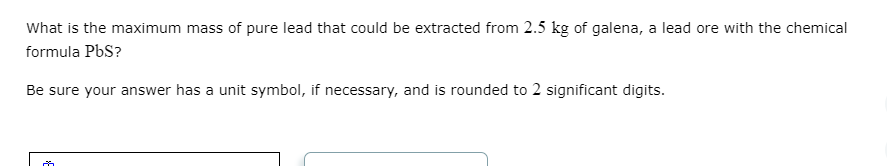
D Try Again Your answer is incorrect. • C3 S₂: Your answer is incorrect. • BP: The name of this compound is not boron phosphide . Be sure you know the correct names of all elements in this compound. Check the Periodic Table if necessary. Fill in the systematic names of the following chemical compounds. Note: for compounds containing hydrogen, you may give the common name instead. molecular formula BI3 C3 S2 BP CS2 H₂O name of compound boron triiodide carbon subsulfide boron phosphide carbon disulfide water What is the maximum mass of pure lead that could be extracted from 2.5 kg of galena, a lead ore with the chemical formula PbS? Be sure your answer has a unit symbol, if necessary, and is rounded to 2 significant digits. m D Try Again Your answer is incorrect. • C3 S₂: Your answer is incorrect. • BP: The name of this compound is not boron phosphide . Be sure you know the correct names of all elements in this compound. Check the Periodic Table if necessary. Fill in the systematic names of the following chemical compounds. Note: for compounds containing hydrogen, you may give the common name instead. molecular formula BI3 C3 S2 BP CS2 H₂O name of compound boron triiodide carbon subsulfide boron phosphide carbon disulfide water What is the maximum mass of pure lead that could be extracted from 2.5 kg of galena, a lead ore with the chemical formula PbS? Be sure your answer has a unit symbol, if necessary, and is rounded to 2 significant digits. m
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Please read the question properly and answer according to that. Please do not copy from other answers because those are wrong. I will upvote you if the answer is correct. 15 K 15 V 100 K Assume for...
-
Please answer and discuss/integrate question of the following three categories: Strengths and Development Areas What are your strongest attributes as a leader? Needs How much personal time do you...
-
please help explain and answer study question number 2 & 3 page discussion questions 1. refer to the fundamental principles governing an audit (see chapter 2@ ). under the responsibili
-
In Figure, a square of edge length 20.0 cm is formed by four spheres of masses m1 = 5.00 g, m2 = 3.00 g, m3 = 1.00 g, and m4 = 5.00 g. In unit-vector notation, what is the net gravitational force...
-
Audit Risk Model. Audit risks for particular accounts and disclosures can be conceptualized in the model: Audit risk (AR) 5 Inherent risk (IR) 3 Control risk (CR) 3 Detection risk (DR). Use this...
-
Costs of quality (CMA, adapted) Costen, Inc., produces cell phone equipment. Jessica Tolmy, Costens president, decided to devote more resources to the improvement of product quality after learning...
-
Reconsider the data from Problem 4. Management has expressed some concern over the life of the project and the impact of possible early termination. As a result, you have developed additional data...
-
Depreciation for Partial PeriodsSL, Act, SYD, and DDB the cost of equipment purchased by Charleston, Inc., on June 1, 2010 is $89,000. It is estimated that the machine will have a $5,000 salvage...
-
A 15 kg box is given a shove to the right on a horizontal, rough surface. After the hand loses contact, the box's acceleration (using standard directions) is a = (-1.2 m/s, 0). (a) What are the...
-
Write a function: string solution (string &S, string &T); that, given two strings S and T consisting of N and M characters, respectively, determines whether string T can be obtained from string S by...
-
How do hospitals determine the quality measures to be used with dashboards, and how are these related to the financial health of the health care organization?
-
A TK 1 , 0 0 0 - face - value bond has a current market price of TK 9 3 5 , an 8 percent coupon rate, and 1 0 years remaining until maturity. Interest payments are made semiannually. What is the...
-
Are you someone who is affected by the hostile media effect or do you know someone who is? How do you react when you see balanced news reports that include political views that you don't like? Are...
-
How do lambda expressions contribute to the principles of declarative programming and functional purity, enabling the specification of computations through function composition and the application of...
-
In the context of software testing and quality assurance, discuss strategies for designing effective test cases and test suites that exercise different branches and conditions within "if" statements,...
-
The cost - volume - profit analysis ( CVP ) is based on the assumption that there are linear relationships among all cost and revenue variables. This linearity suggests practical and conceptual...
-
A coffee franchise owner opened a local coffee shop, called MUG, in one of the suburbs of New York City. The coffee shop estimates it uses 3,000 pounds of coffee annually. The manager of MUG has to...
-
The following cost information was provided to you for analysis: September 12,000 Units Produced Costs: TIC TAC TOE TING August 10,000 P80,000 70.000 60.000 50,000 How much is the fixed cost per...
-
Ann hires a nanny to watch her two children while she works at a local hospital. She pays the 19-year-old nanny $125 per week for 48 weeks during the current year. a. What is the employer's portion...
-
Charlie's Green Lawn Care is a cash basis taxpayer. Charlie Adame, the sole proprietor, is considering delaying some of his December 2012 customer billings for lawn care into the next year. In...
-
For each of the following situations, indicate whether the taxpayer(s) is (are) required to file a tax return for 2012. Explain your answer. a. Helen is a single taxpayer with interest income in 2012...
-
Acne Medication and Gastrointestinal Injury Since 1999, Kamie Kendall had experienced ongoing abdominal pain, and she was eventually diagnosed with ulcerative colitis and irritable bowel syndrome...
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
CMS is a claims processing company in Mobile, Alabama. Chastity Jones, a black woman, completed an online employment application for a customer service position with CMS. Jones interviewed with a...

Study smarter with the SolutionInn App


