Which of the following relationships is correct at constant T and P? A. AG is proportional...
Fantastic news! We've Found the answer you've been seeking!
Question:
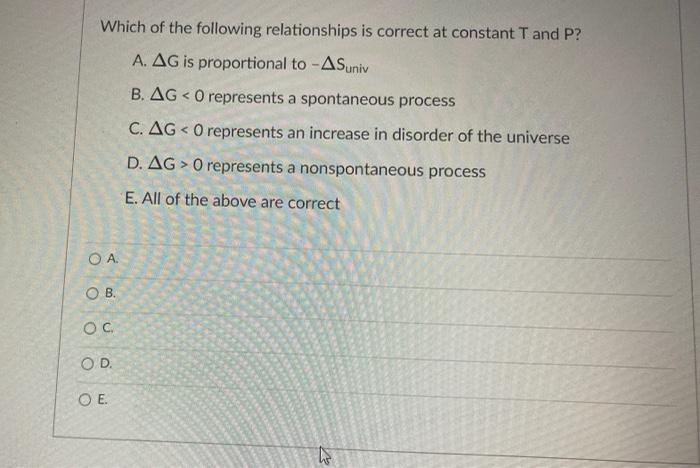

Transcribed Image Text:
Which of the following relationships is correct at constant T and P? A. AG is proportional to -ASuniv B. AG < 0 represents a spontaneous process C. AG< 0 represents an increase in disorder of the universe D. AG> 0 represents a nonspontaneous process E. All of the above are correct OA. OB. OC OD. O E. If a reaction is exothermic and results in decreased entropy, what is true about the reaction? A. This reaction will be spontaneous only at low temperatures. B. This reaction will be spontaneous at all temperatures. C. This reaction will be nonspontaneous at all temperatures. D. This reaction will be nonspontaneous only at low temperatures. E. It is not possible to determine without more information. OA. OB. OC OD. OE Which of the following relationships is correct at constant T and P? A. AG is proportional to -ASuniv B. AG < 0 represents a spontaneous process C. AG< 0 represents an increase in disorder of the universe D. AG> 0 represents a nonspontaneous process E. All of the above are correct OA. OB. OC OD. O E. If a reaction is exothermic and results in decreased entropy, what is true about the reaction? A. This reaction will be spontaneous only at low temperatures. B. This reaction will be spontaneous at all temperatures. C. This reaction will be nonspontaneous at all temperatures. D. This reaction will be nonspontaneous only at low temperatures. E. It is not possible to determine without more information. OA. OB. OC OD. OE
Expert Answer:
Answer rating: 100% (QA)
Answers A AG is proportional to ASuniv B AGO represents a spontaneous process D AG 0 represents a nonspontaneous process A This reaction will be spontaneous only at low temperatures Explanation 1 Answ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Please read the question properly and answer according to that. Please do not copy from other answers because those are wrong. I will upvote you if the answer is correct. 15 K 15 V 100 K Assume for...
-
A hotel uses a booking system that needs to store, per booking, the room num- ber, the duration of the booking (start date, end date), a reference number for the guest, and finally the reference...
-
Briefly explain your answers Fig.1 1. Assuming the monopoly pictured above is a profit maximizing monopoly, what price will the monopoly charge for its output? 2. The profit maximizing monopoly will...
-
What are the primary reasons for separating operational tasks?
-
f(x) = log 3 (x + 5) and g(x) = log 3 (x - 1). (a) Solve f(x) = 2. What point is on the graph of f? (b) Solve g(x) = 3. What point is on the graph of g? (c) Solve f(x) = g(x). Do the graphs of f and...
-
The adjusted trial balance for Spruce Up Catering, Inc., is presented next. Prepare the income statement, statement of retained earnings, and balance sheet for Spruce Up Catering, Inc., for the month...
-
Gerald Pat Arrington was diagnosed with a brain tumor. At the time of the diagnosis, he was married to Brenda Arrington, but they were separated pending their divorce. Brenda and Pat had no children,...
-
You have just been hired as a financial analyst for Lydex Company, a manufacturer of safety helmets. Your boss has asked you to perform a comprehensive analysis of the company's financial statements,...
-
When Howard Foth died in the current year, in addition to his $10,745,000 of certificates of deposit and investment property worth $200,000, the following facts were disclosed by the executor: a. In...
-
A client calls in to sell their entire long position (300 shares) of AAPL and wanted you to walk them through the process. While going through BPROS you notice they are also short 3 calls contracts...
-
A 4.33 kilogram object is 286.75 meters above the ground. What is the object potential energy in joules at this height? Round up to two decimal places
-
If a group of angel investors invests $7,750,000 now, they will receive 27% of the exit value given the expected rate of return is 33% on their investment. What is the firm's exit value at the end of...
-
Bandar Industries manufactures sporting equipment. One of the company's products is a football helmet that requires special plastic. During the quarter ending June 30, the company manufactured 3,100...
-
The management of International Cookwares is looking at the effectiveness of its stamping machine. Here is last week's data for the machine:- Good units (defect free) produced: 12,000 Machine down...
-
11.5 ABC Manufacturing has the following results for the year ended December 31, 2023: Total Revenue $12,500,000 Net Income 10% Total Assets $80,000,000 Total Liabilities $30,000,000 Tax rate 15%...
-
Ahmad encounters a risk that he thinks must be brought to the attention of his superiors or at the very least, to the attention of another relevant part of the organization, so that they can deal...
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
Calculate the concentration of Pb2+ in each of the following.
-
Consider the hypothetical reaction B E + F which is assumed to occur by the mechanism Where B* represents a B molecule with enough energy to surmount the reaction energy bar70. Consider the following...
-
For each of the following pairs of elements, (C and N) (Ar and Br) (Mg and K) (F and Cl) pick the one with a. The more favorable (exothermic) electron affinity b. The higher ionization energy c. The...
-
What is the average rate of return for the values that follow? 0.90, 1.1, 1.2
-
What is the average rate of return for the values that follow? 0.80, 2.0, 5.0
-
State whether the data are symmetrical, skewed to the left, or skewed to the right. 16; 17; 19; 22; 22; 22; 22; 22; 23

Study smarter with the SolutionInn App


