A thermodynamic cycle in a vapor power plant has four states and four processes. States can...
Fantastic news! We've Found the answer you've been seeking!
Question:
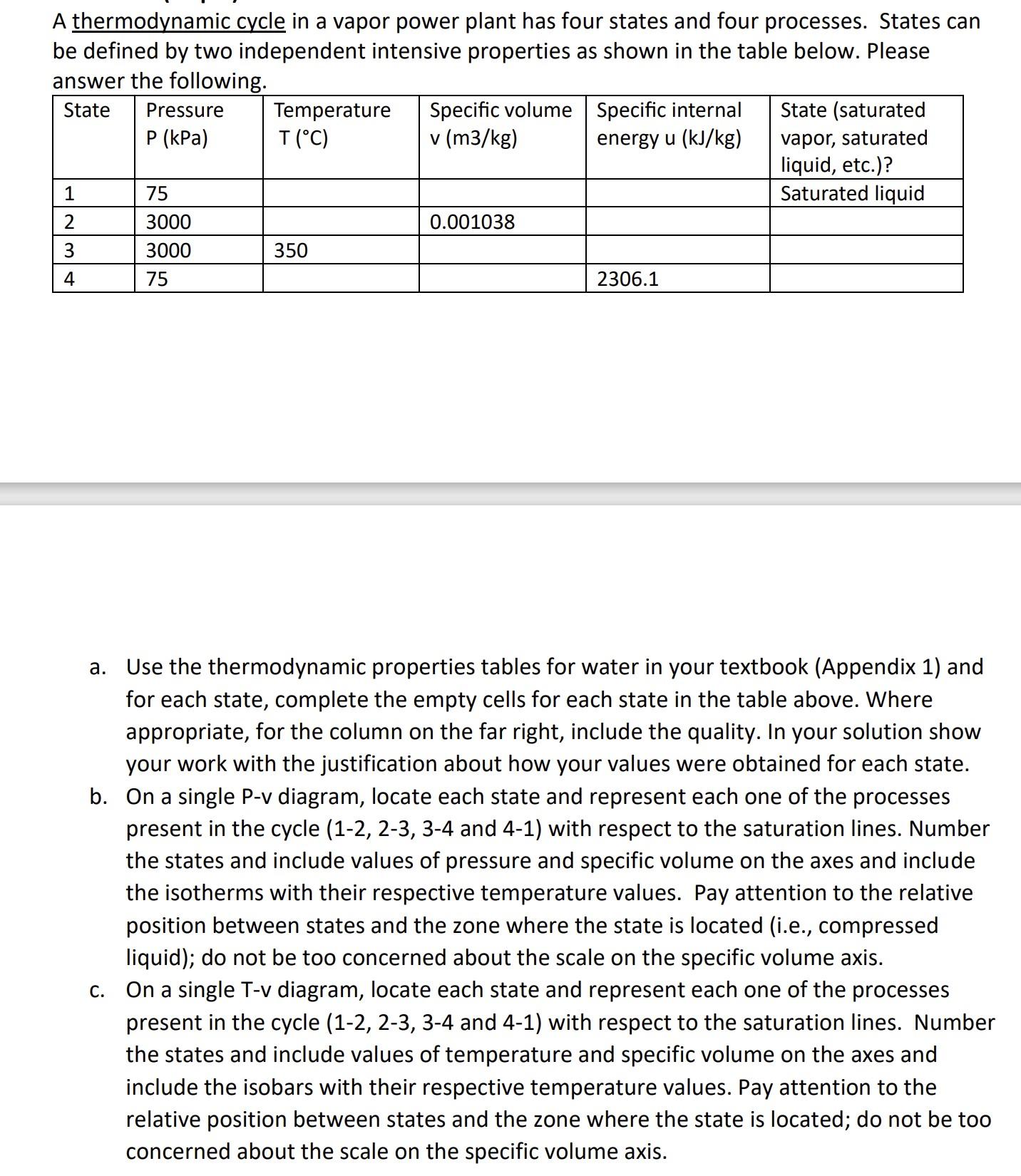
Transcribed Image Text:
A thermodynamic cycle in a vapor power plant has four states and four processes. States can be defined by two independent intensive properties as shown in the table below. Please answer the following. State Pressure P (kPa) 1 2 34 75 3000 3000 75 a. Temperature T (°C) 350 Specific volume v (m3/kg) 0.001038 Specific internal energy u (kJ/kg) 2306.1 State (saturated vapor, saturated liquid, etc.)? Saturated liquid Use the thermodynamic properties tables for water in your textbook (Appendix 1) and for each state, complete the empty cells for each state in the table above. Where appropriate, for the column on the far right, include the quality. In your solution show your work with the justification about how your values were obtained for each state. b. On a single P-v diagram, locate each state and represent each one of the processes present in the cycle (1-2, 2-3, 3-4 and 4-1) with respect to the saturation lines. Number the states and include values of pressure and specific volume on the axes and include the isotherms with their respective temperature values. Pay attention to the relative position between states and the zone where the state is located (i.e., compressed liquid); do not be too concerned about the scale on the specific volume axis. c. On a single T-v diagram, locate each state and represent each one of the processes present in the cycle (1-2, 2-3, 3-4 and 4-1) with respect to the saturation lines. Number the states and include values of temperature and specific volume on the axes and include the isobars with their respective temperature values. Pay attention to the relative position between states and the zone where the state is located; do not be too concerned about the scale on the specific volume axis. A thermodynamic cycle in a vapor power plant has four states and four processes. States can be defined by two independent intensive properties as shown in the table below. Please answer the following. State Pressure P (kPa) 1 2 34 75 3000 3000 75 a. Temperature T (°C) 350 Specific volume v (m3/kg) 0.001038 Specific internal energy u (kJ/kg) 2306.1 State (saturated vapor, saturated liquid, etc.)? Saturated liquid Use the thermodynamic properties tables for water in your textbook (Appendix 1) and for each state, complete the empty cells for each state in the table above. Where appropriate, for the column on the far right, include the quality. In your solution show your work with the justification about how your values were obtained for each state. b. On a single P-v diagram, locate each state and represent each one of the processes present in the cycle (1-2, 2-3, 3-4 and 4-1) with respect to the saturation lines. Number the states and include values of pressure and specific volume on the axes and include the isotherms with their respective temperature values. Pay attention to the relative position between states and the zone where the state is located (i.e., compressed liquid); do not be too concerned about the scale on the specific volume axis. c. On a single T-v diagram, locate each state and represent each one of the processes present in the cycle (1-2, 2-3, 3-4 and 4-1) with respect to the saturation lines. Number the states and include values of temperature and specific volume on the axes and include the isobars with their respective temperature values. Pay attention to the relative position between states and the zone where the state is located; do not be too concerned about the scale on the specific volume axis.
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these law questions
-
Please pick a side. Do you see the era of womens protective labor legislation as a positive or negative when it comes to advancing womens position in paid employment? Whichever position you took in...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Goals of this unit: 1. construct cogent, logical, effective, and ethical arguments in writing; 2. compose texts that effectively employ the features of a given genre; 3. identify reliable and...
-
Find the smallest positive angle and the smallest negative angle (numerically) coterminal with but not equal to the given angle. 47.0
-
A pressure cooker (closed tank) contains water at 200 F with the liquid volume being 1/10 of the vapor volume. It is heated until the pressure reaches 300 lbf/in 2. Find the final temperature. Has...
-
Complete the following, determining the appropriate uncertainty: ( a ) ( 2 . 7 0 \ pm 0 . 0 5 cm ) 2 ( b ) ( 2 . 7 0 \ pm 0 . 0 7 cm ) 3 ( c ) ( 1 2 . 0 2 \ pm 0 . 0 8 cm ) 4 ( d ) ( 2 0 . 6 ...
-
Briefly describe the role of technology in conceptual, logical, and physical modeling.
-
Refer to the Professional Judgment in Context feature "Deficiencies in Auditing Debt Obligations and Stockholders' Equity Accounts: Insights from SEC Releases" and review the panel related to...
-
Now that we know about data, we continue our journey with 2 fundamental descriptive statistics that help us understand fundamental components of a data set. In this week's discussion, we are going to...
-
Hotel DelRay is located at the heart of the city of Brussels, in Belgium. Brussels is a major hub for international politics, a home for several international organizations and diplomats, and a...
-
Assume that a national restaurant firm called BBQ builds 10 new restaurants at a cost of $1 million per restaurant. It outfits each restaurant with an additional $200,000 of equipment and...
-
A clerk is maintaining three different files containing job applications submitted for three different positions currently open in the firm at which the clerk is employed. One file contains two...
-
The miles per gallon attained by purchasers of a line of pickup trucks manufactured in Detroit are outcomes of a random variable with a mean of 17 miles per gallon and a standard deviation of .25...
-
This year, FCF, Inc., has earnings before interest and taxes of $10,300,000, depreciation expenses of $1,200,000, capital expenditures of $1,600,000, and has increased its net working capital by...
-
You purchased a machine for $1.09 million three years ago and have been applying straight-line depreciation to zero for a seven-year life. Your tax rate is 21%. If you sell the machine today (after...
-
Karl Stick is president of Stock Company. He also owns 100 percent of its stock. Karl's salary is $220,000. At the end of the year, Karl was paid a bonus of $100,000 because the firm had a good year....
-
x and y in the space loo such that 11 sel|| = ||y|| = 1 and || 2+yll = 2. ST there enist two vector they are linearly independent,
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
Professor Patricia (Patty) Pate is retired from the PalmSprings Culinary Arts Academy (PSCAA). She is a single taxpayer and is 68 years old. Patty lives at 98 Colander Street, Henderson, NV 89052....
-
John Fuji (age 37) moved from California to Washington in December 2011. He lives at 468 Cameo Street, Yakima, WA 98901. John's Social Security number is 571-78-5974 and he is single. His earnings...
-
Leslie and Leon Lazo are married and file a joint return for 2012. Leslie's Social Security number is 466-47-3311 and Leon's is 467-74-4451. They live at 143 Snapdragon Drive, Reno, NV 82102. For...
-
In a large office building, there is a woman dead in her locked office, sitting at her desk. Except for the desk, chair, and contents on the desk, the office is empty and the air conditioner is...
-
How many birthdays does the average person have?
-
There are 5 different color houses, occupied by people of 5 different nationalities, who smoke 5 different cigar brands, drink 5 different types of alcohol, and have 5 different pets. The houses are...

Study smarter with the SolutionInn App


